AbstractObjectiveVestibular schwannoma (VS) is a benign, slow-growing tumor originating from Schwann cells of the cranial nerve. Many studies have reported short-term follow-ups of patients with VS undergoing gamma knife radiosurgery (GKR). This study analyzed factors affecting the treatment and prognosis of patients over a 10-year period who underwent GKR at a single center by 1 surgeon.
MethodsBetween 1990 and 2003, we used GKR as a primary treatment or after prior surgical tumor removal for patients with VS. During this period, a single surgeon in 1 center treated more than 700 patients. Of these, 182 patients available for long-term follow-up via the outpatient department were included in the study. Among them, 156 patients were available for radiological imaging follow-up; patients who were not available for radiological follow-up were excluded. Initial tumor volume, initial prescription dose, prior surgery, neurological symptom improvement, and tumor control by individual status were assessed to analyze the factors associated with treatment outcomes.
ConclusionAt more than 10-years’ follow-up, GKR for VS was effective for tumor control in 91.0% of cases and there were few complications associated with GKR. With improvements in the technology and precision of gamma knife systems, planning programs, and other treatment protocols such as fraction and low marginal dose, we should continuously study following results. Our study results suggest the need for more effective methods for tumor control and fewer neurological and GKR-associated complications.
INTRODUCTIONVestibular schwannoma (VS) is a benign, slow-growing tumor that originates from the Schwann cells of the cranial nerve. Several advancements in high-quality neuroimaging techniques and knowledge of VS have enabled the relatively early diagnosis and treatment of patients with VS. In addition, advancements in precise methods and accurate protocols for stereotactic radiosurgery technique have also occurred. Gamma knife radiosurgery (GKR) is currently used as a primary treatment method for VS of small size or as a secondary treatment for post-operative residual tumors.
Reports have indicated the short-term clinical outcomes of patients with VS treated with GKR; however, few reports include long-term follow-up data. Some reports have indicated side effects or treatment failure of GKR such as tumor progression and post-radiation edematous changes. Therefore, studies to evaluate long-term outcomes are necessary.
This large-scale study aimed to retrospectively evaluate the definite clinical and radiological outcomes after treatment among GKR cases performed by a single surgeon at a single institution.
MATERIALS AND METHODSAt Asan Medical Center, more than 700 patients were treated with GKR for VS during a >20-year period. Among them, 182 patients with more than 10 years of treatment follow-up were available and included in the present study. The clinical data of these patients are summarized in Table 1. The mean patient age was 51 (range, 17-79) years; 55 were male (30.2%) and 127 were female (69.8%). A total of 57 patients (31.3%) had undergone prior surgery.
The evaluation of patient status was based on cranial nerve dysfunction [3,9,11], which indicated that 49 patients had 5th nerve deficits and 42 patients had 7th nerve palsy. Among patients who had 8th nerve symptoms, 50 had hearing disturbance and 96 had deafness; in addition, 45 and 29 patients had tinnitus and dizziness, respectively. The mean tumor volume was 5,206.4 (28-28, 380) mm3 and the mean dose to the tumor margin was 12.83 (11-17) Gy. Some exceptional overdose cases or indication of initial tumor volume were observed in the early period.
GKR was performed using Leksell frame for stereotactic imaging under local anesthesia and mild sedation was provided if needed. On May 8, 1990, GK B-type and KULA planning systems (Kula system; Elekta Instrument AB, Stockholm, Sweden) were introduced at our center, and GKR was started. On November 22, 1999, an image-based GammaPlan was applied. C-type GK was introduced in 2006 and a Gamma Knife Perfexion (Elekta Instrument AB) was introduced in 2010 in sequence. Lastly, a GK ICON (Elekta Instrument AB) was introduced as of March 8, 2017, and fractions were made possible by using masks [1,8,15]. Detailed GKR were planned, including tumor targeting, volume evaluation, and dose-planning based on highresolution GKR magnetic resonance (MR) (thin section) and the radiation margin was carefully established considering the adjacent critical structures such as the pontomedullary junction and cranial nerve route. After treatment, clinical and radiological follow-ups were scheduled every 6 months or 1 year after treatment. Clinical follow-up focused on trigeminal, facial, and vestibulocochlear nerve function preservation or aggravation was conducted. If necessary, visits to the outpatient department (OPD) were required for patients whose general condition or neurologic status had changed. Additionally, the mortality statistics of VS after GKR were investigated by classification of the patients into 2 age groups (<60 and >60 years).
RESULTSIn this study, radiological and clinical follow-up was possible in 156 (85.7%) patients and the mean follow-up period calculated from the initial OPD follow-up date to last the OPD follow-up date was 84.52 (9-145) months. During these period, 129 patients were alive and 27 had died; among these, 2 patients died of complications related to increased tumor volume and post-operation-related aspiration pneumonia and pulmonary embolism, 3 patients died of underlying malignancy, and the remaining three patients each died of subarachnoid hemorrhage, trauma, and other major stroke events (Table 2).
During the follow-up period, the cause of death was unknown in 19 of the 27 (17.3%) mortality cases. Increased tumor volume and subsequent operation-related death were noted in 2 patients. Among them, 15 patients were >60 years of age at the time of treatment. VS is a benign tumor and, as expected, GKR had no effect on mortality, regardless of age group. There was also no significant difference between the average mortality rate in normal individuals >60 years of age. The mean age at death was 67.7 years and the average duration of life was 9.7 years. Hydrocephalus was diagnosed in 10 patients, 6 of whom died. There was no significant difference in the life span between patients with hydrocephalus (mean, 9.3 years) and others (mean, 9.7 years).
In follow-up MR imaging (MRI) after GKR, increased tumor volume was noted in 14 (9.0%) patients and tumor control (stable or decrease) in 142 (91.0%) (Table 3). For patients with enlarged tumors, the average follow-up duration was 21.4 months (range, 5-136 months). Among patients with increased tumor size, 5 died: One patient died of complications due to rectal cancer (pulmonary embolism); the other 3 patients underwent follow-up for 5, 6, and 7 years respectively, and 1 patient died of unknown cause; lastly, 1 patient underwent resection surgery due to an increase in tumor size 5 months after GKR treatment and died 2 years post-GKR.
The estimated volume of the enhanced portion showing gradual loss was observed (Fig. 1). As shown in the figure, VS located at the left cerebellopontine angle was observed in the initial axial T1-weighted images. The patient underwent GKR (12 Gy margin dose at the 50% isodose level) and follow-up. The initial brain tumor volume was approximately 2.57 cc and the maximal dimension was about 20 mm. At the first follow-up brain MRI performed 13 months after GKR, a typical decrease of the well-enhanced mass was observed on the acquired axial T1-weighted enhanced image. At additional follow-up brain MRI performed at 42 months, gradual decreases in the tumor size and volume were observed. A stable status of the treated tumor was achieved based on a decrease in tumor size at MRI performed 12 and 14 years later.
In statistical analysis, pre-GKR tumor volume was a significant factor in cases with increased tumor size versus those with controlled tumor size [1,7]. In the evaluation of data from our center, the patients were classified into 4 groups by tumor size. In the first group, among 39 patients, 2 cases had increased tumor size. Increased tumor size was observed in 1, 5, and 6 cases in the second, third, and fourth groups, respectively. Statistical significance (p=0.049) of tumor-control rate for post-GKR was achieved (Table 3).
A total of 57 patients had prior surgery, including 2 cases of VP shunt surgery with obstructive hydrocephalus. Among them, 23 cases had prescribed doses of more than 14 Gy. At the beginning stages of gamma knife modality, the treatment methods, plans, and prescription doses were not as elaborate or detailed as those currently in use. In 45 cases, the residual tumor size was >13,000 mm3. With regard to treatment with GKR after prior surgery, 50 cases achieved stable or decreased tumor size.
In cases of large initial tumor volume prior to the requirement for decompressive surgery [4,12], there was no significant difference in neurologic outcomes between the groups of primary GKR and GKR after prior surgery. Differences in tumor size and neurologic symptom were compared between the prior surgery and primary GKR groups. Based on the patients’ subjective symptoms, neurologic symptoms were categorized as improved, stationary, or worsened. Analysis of data from our center revealed no significant relationship in tumor size changes between the 2 groups.
According to the prescribed dose, the patients were classified into 2 groups; namely, up to 13 Gy and over 14 Gy. No significant differences were observed in the improvement of neurological symptoms according to the dose.
The results of the clinical and radiological follow-ups are shown in Table 4. Improvements in hearing function and tinnitus were rarely checked. However, GKR of VS had significant effects in terms of preventing the deterioration of neurologic function.
In addition, the effect of GKR treatment was analyzed according to the pre-treatment neurological symptoms, prescription dose, and age according to the tumor size. Comparisons of GKR treatment outcomes according to initial tumor size indicated a positive correlation between tumor volume control and smaller initial tumor volume (Table 3). There was no significant difference in the changes in neurological symptoms according to initial tumor volume and patient age (Table 5, 6).
Previously, dose prescription and planning were not as elaborate as those in current practice and treatment with higher doses was administered to the groups of patients with prior surgical reduction or with large initial tumor volumes. The patients were divided into 2 groups according to the prescribed dose (up to 13 Gy or 14 Gy or greater). Comparison of neurologic symptoms between the subgroups revealed no statistically significant differences in the dose level or neurologic improvement (Table 7).
DISCUSSIONVS is a benign tumor that is generally not life-threatening. Nevertheless, VS can cause neurologic dysfunction involving the 5th, 7th, and 8th cranial nerves, such as facial palsy and hearing dysfunction. Typically, VS is not fast-growing; however, as tumor size increases, neurologic deterioration and other mass effects such as hydrocephalus or brainstem compression may occur. Therefore, adequate treatment of VS is important for a good quality of life and prognosis.
The tumor control rate (91.0%) was relatively low because the indication regarding tumor volume was larger in the early 1990’s for GKR compared to the current indication. At that time, clinicians had to treat patients with large remaining tumors with post-surgical reduction. Several factors may contribute to the success rate. Accurate GKR software programs to plan treatment margins, dose, and fractions based on the radiological workup of patients have been developed. Moreover, MR resolution is in a range that is specific and suitable for GKR [1,6,8,15]. In our study, increased tumor size and deterioration of neurologic symptoms was generally associated with initial tumor size. A previous study reported that 12 patients with 14 cases of tumor progression with increased size after GKR were non-responders and that the initial tumor size was larger than 150 mm [3,14].
In the retrospective long-term follow-up results of GKR in our study, the occurrence rate of facial palsy was high due to high tumor marginal dose compared to that with low marginal dose GKR performed recently. As a result, there is a lower risk of neurological deterioration and other complications due to peritumoral swelling after treatment.
The results of the analysis of the GKR data in our study demonstrated a relationship between tumor size and treatment outcomes. As the size increased, the tumors were considered unsuitable for GKR treatment. In these cases, other treatment options such as tumor resection should be considered first. With advances in radiosurgery strategy, the indications have become more precise. Previous data have revealed that larger initial tumor volume and diameter were not sufficiently considered and in cases with larger initial tumor volume, significantly poor outcomes were likely. Clinicians should select suitable candidates for GKR treatment to ensure good treatment outcomes. In agreement with a study conducted at another center, we considered GKR candidates to be those patients with VS size <3 cm in diameter and 150 mm3 in initial volume.
Recently, we performed fractionated radiosurgery with a GKR ICON, which allowed us to protect and save important neighboring structures such as the cranial nerve; thus, a better effect could be expected. Studies including further analysis of fraction and follow-up in VS patients with small tumor volume and hearing preservation are required to develop approaches that preserve neurologic function in these patients.
Our previous report indicated the effectiveness of GKR in patients at our center with VS with cyst components [5]; the analysis was based on factors such as the cyst volume ratio, cyst type, and past resection history or prescription dose. In the case of VS with cystic portions, the neurological deterioration was faster than that in VS with solid components and the prognosis was not easily analyzed [13].
The biomechanism of post-treatment edema is unclear; however, extravasation of protein due to changes in vascular permeability and transient GKR-induced swelling which results in increased protein levels may lead to poor prognosis due to the progression of the cystic component from increased osmotic pressure.
In our present study, the prognosis was poor in patients with large cysts. While aspiration may be considered for metastatic brain tumors at other locations, this procedure is difficult in the case of VS due to its location. In these cases, additional GKR for residual tumor after surgical decompression can be considered.
A recent case of unruptured aneurysm was treated with coil embolization and continuous antiplatelet therapy (aspirin) and GKR for VS was performed 3 months later. The patient was discharged without any other specific neurologic deficits. At 1 month after GKR, the patient experienced discomfort due to pain in the left ear and underwent MRI. The images showed intratumoral hemorrhage in the Gradient echo sequence. The patient found the condition to be tolerable and had no other specific deficits. Thus, considering the presence of underlying cerebrovascular diseases or other antiplatelet treatments, clinicians should pay careful attention to tumor bleeding.
Two cases of additional treatment for recurred VS including those of primary surgery and GKR were reported [2]; in the first case, the patient underwent additional decompressive surgery due to the increased size of the tumor cyst and thereafter achieved stable status. In the other case, the patient underwent additional GKR after 15 years, with subsequent tumor disappearance [12]. With regard to the treatment of intracanalicular tumors with small volume in patients with intact hearing, recent studies have recommended a wait-and-see approach and only conservative treatment with frequent follow-up intervals. Many cases may result in neurologic deterioration; therefore, tumor control through early GKR is necessary in such cases [10].
CONCLUSIONAt more than 10 years’ follow-up, GKR for VS was effective for tumor control (91.0%) and showed a low rate of associated complications, which indicated that GKR was an effective treatment in these patients. Low marginal dose GKR was performed, which resulted in decreased complication rates. Low marginal dose GKR enabled long-term control of the tumor status and lower complications including neurologic deterioration and other post-GKR-related events. The delicate and precise features of advanced stereotactic MR systems can contribute to procedure planning and successful treatment of VS. Studies related to fractionated GKR for VS with continuous follow-up are needed to improve the understanding of its effects and results.
Fig. 1.Magnetic resonance images obtained in a 40-year-old woman with vestibular schwannoma at left the cerebellopontine angle. (A) Axial T1-weighted images. Gamma knife radiosurgery (GKR) was conducted with 12 Gy margin dose at a 50% isodose level. The original tumor volume was 2.57 cc and the maximal dimension was about 20 mm (arrow). (B) At 13 months after GKR, a typical decrease in the well-enhanced mass was noted. (C) The tumor volume showed a gradual decrease to 17mm at 42 months. (D) A residual lesion approximately 15 mm in size was observed. (E, F) Decreases in the tumor size and volume of 1.37 and 0.67 cc, respectively, were observed, indicating a relatively stable status at 12 and 14 years. 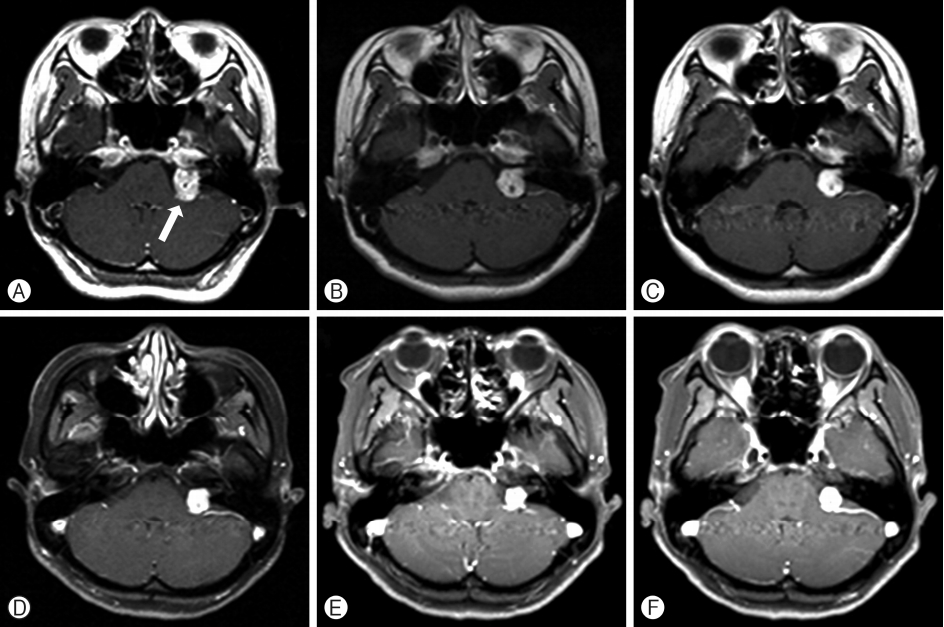
Table 1.Baseline characteristics and treatment parameters Table 2.Follow-up mortality after gamma knife radiosurgery Table 3.Volume control after gamma knife radiosurgery according to initial tumor volume
Table 4.Clinical and radiologic follow-up results of gamma knife radiosurgery (n=156)
Table 5.Clinical follow-up results of gamma knife radiosurgery according to initial tumor volume (n=156)
Table 6.Clinical follow-up results of gamma knife radiosurgery according to age (n=156) Table 7.Clinical follow-up results of gamma knife radiosurgery according to initial prescription dose (n=156)
REFERENCES1. Casentini L, Fornezza U, Perini Z, Perissinotto E, Colombo F: Multisession stereotactic radiosurgery for large vestibular schwannomas. J Neurosurg 122:818-824, 2015
2. Fu VX, Verheul JB, Beute GN, Leenstra S, Kunst HPM, Mulder JJS, et al.: Retreatment of vestibular schwannoma with Gamma Knife radiosurgery: clinical outcome, tumor control, and review of literature. J Neurosurg 129:137-145, 2018
3. Hasegawa T, Kato T, Yamamoto T, Naito T, Kato N, Torii J, et al.: Long-term hearing outcomes after gamma knife surgery in patients with vestibular schwannoma with hearing preservation: evaluation in 92 patients with serial audiograms. J Neurooncol 138:283-290, 2018
4. Huang CW, Tu HT, Chuang CY, Chang CS, Chou HH, Lee MT, et al.: Gamma Knife radiosurgery for large vestibular schwannomas greater than 3 cm in diameter. J Neurosurg 128:1380-1387, 2018
5. Lee JM, Kwon DH, Kim CJ, Kim JH: Treatment outcome of gamma knife radiosurgery of vestibular schwannomas with cystic component. Nerve 2:1-4, 2016
6. Lo A, Ayre G, Ma R, Hsu F, Akagami R, McKenzie M, et al.: Population-based study of stereotactic radiosurgery or fractionated stereotactic radiation therapy for vestibular schwannoma: Long-term outcomes and toxicities. Int J Radiat Oncol Biol Phys 100:443-451, 2018
7. Milligan BD, Pollock BE, Foote RL, Link MJ: Long-term tumor control and cranial nerve outcomes following gamma knife surgery for larger-volume vestibular schwannomas. J Neurosurg 116:598-604, 2012
8. Nguyen T, Duong C, Sheppard JP, Lee SJ, Kishan AU, Lee P, et al.: Hypo-fractionated stereotactic radiotherapy of five fractions with linear accelerator for vestibular schwannomas: A systematic review and meta-analysis. Clin Neurol Neurosurg 166:116-123, 2018
9. Putz F, Müller J, Wimmer C, Goerig N, Knippen S, Iro H, et al.: Stereotactic radiotherapy of vestibular schwannoma: Hearing preservation, vestibular function, and local control following primary and salvage radiotherapy. Strahlenther Onkol 193:200-212, 2017
10. Régis J, Carron R, Park MC, Soumare O, Delsanti C, Thomassin JM, et al.: Wait-and-see strategy compared with proactive Gamma Knife surgery in patients with intracanalicular vestibular schwannomas: clinical article. J Neurosurg 119 Suppl:105-111, 2013
11. Santa Maria PL, Shi Y, Gurgel RK, Corrales CE, Soltys SG, Santa Maria C, et al.: Long-term hearing outcomes following stereotactic radiosurgery in vestibular schwannoma patients-a retrospective cohort study. Neurosurgery [epub ahead of print, 2018. doi: 10.1093/neuros/nyy407]
12. Shimansky VN, Odamanov DA, Ryzhova MV, Tanyashin SV, Golanov AV, Shevchenko KV, et al.: Surgical approach to resection of vestibular schwannomas following stereotactic radiological treatment. Surgical outcomes and morphological changes in tumors after radiotherapy. Zh Vopr Neirokhir Im N N Burdenko 82:38-52, 2018
13. Shirato H, Sakamoto T, Takeichi N, Aoyama H, Suzuki K, Kagei K, et al.: Fractionated stereotactic radiotherapy for vestibular schwannoma (VS): comparison between cystic-type and solidtype VS. Int J Radiat Oncol Biol Phys 48:1395-1401, 2000
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||