INTRODUCTION
Meningiomas are the most common intradural extramedullary tumors, with more than 75% of them occurring in women, mainly in the thoracic spine. The diagnosis of spinal meningiomas is not very difficult based on radiologic findings and typical locations. Meningiomas are usually benign, slow-growing tumors that derive from arachnoid cap cells in the dura around the dentate ligament of the spinal cord. In most cases, they are located on the side of the spinal cord [11]. The World Health Organization classifies these tumors into Grade I (benign), Grade II (atypical), and Grade III (anaplastic) meningioma. Grade I meningioma is a slowly growing tumor [3] divided into 9 subtypes: meningothelial, fibrous, transitional, psammomatous, angiomatous, microcystic, secretory, lymphoplasmacytic, and metaplastic [14]. Among them, spinal metaplastic meningiomas are very rare. The metaplastic subtype is defined as a tumor containing focal or widespread mesenchymal components including osseous, cartilaginous, lipomatous, myxoid or xanthomatous tissue, singly or in combinations [14]. We report a rare case of confirmed metaplastic meningioma in the thoracic spine in the male.
CASE REPORT
The study was approved by the Institutional Review Board (IRB no. DMC 2019-04-006), informed consent was not needed.
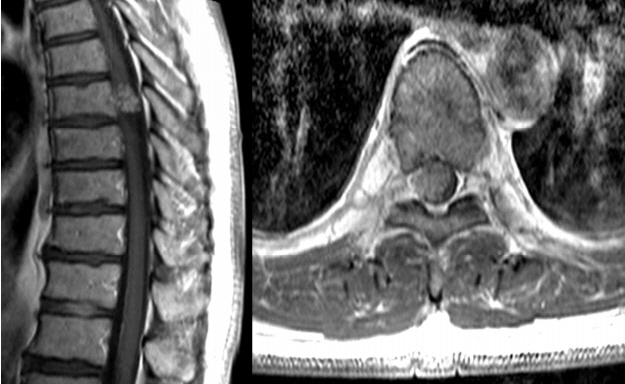
This is a study on a 59-year-old man with thoracic spinal metaplastic meningioma. The man presented with progressively worsening weakness, radiating pain and paresthesia of the right leg. On the past history he received L4/5 fusion due to spinal stenosis and he had hyperesthesia and dysesthesia on light touch and weakness GIII on the right leg. However he did not have symptoms related to bladder or anal sphincter dysfunction. On spinal magnetic resonance imaging (MRI) examination, it showed a 14×11×15 mm sized intradural extramedullary enhancing mass in the posterior epidural space of the right side at the T7 level. The mass caused compression of the spinal cord. T1-weighted images (WIs) with contrast enhancement showed heterogenous enhancement of the mass. The spinal mass showed intermediate signal intensity on T1- and T2-WIs (Fig. 1). Lower extremity electromyography/nerve conduction velocity findings were suggestive of left lumbosacral radiculopathies.
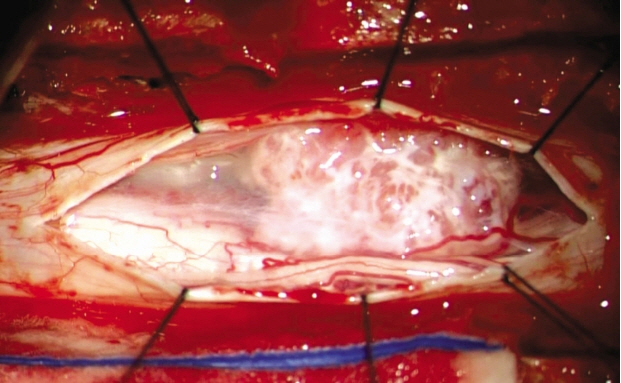
The patient received a mass excision after laminoplasty on the T6 and T7. Microscopic examination of the intradural space mass revealed an intradural calcified multi-lobulated mass with tight adhesion to the thoracic cord and right dura. In addition, there were several rootlets inserted into the calcified tumor. The tumor was a fragment of pinkish red hard tissue with marked ossification (Fig. 2).
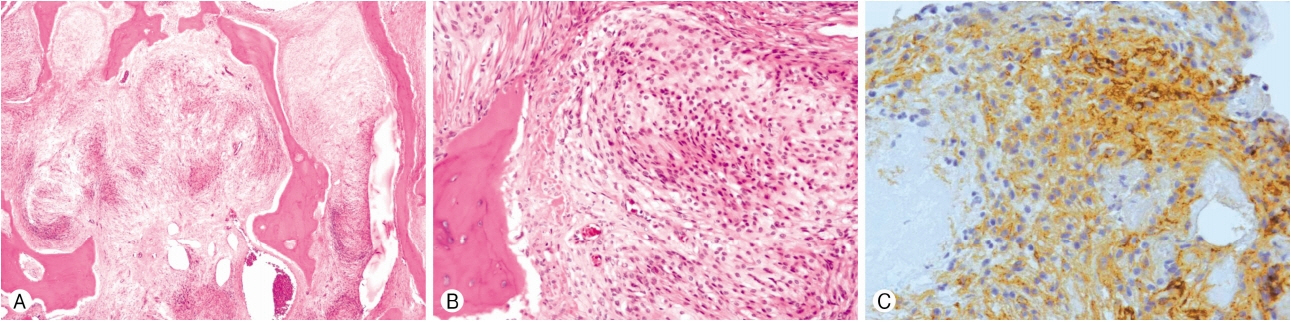
Histologically, the tumor consists of a meningothelial component and osseous tissue (hematoxylin and eosin [H&E], ×40) and the tumor cells show plump, elongated cells with whorl formations (H&E, ×200). The immunohistochemical stain was positive for epithelial membrane antigen (×200) and a Ki 67 antibody was applied but no positive staining was present. The final histological diagnosis was metaplastic meningioma (Fig. 3).
Immediately after surgery, his motor power was grade I of the right leg on the day of surgery. The tumor was removed, but we suspected due to nerve stretching damage after removal of a tightly attached tumor with nerve root. We administered intravenous steroid postoperatively. The next day, his motor power was recovered to Grade III, and Grade IV after active rehabilitation.
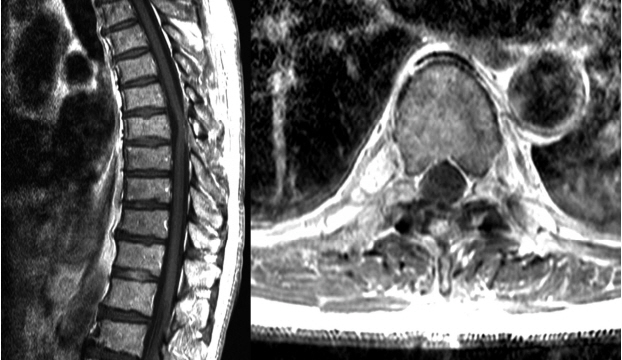
No evidence of tumor recurrence was shown on a five-month follow up MRI (Fig. 4).
DISCUSSION
Spinal meningiomas represent a minority of all meningiomas, approximately 12% [2] and about 25% to 46% of total spinal cord tumors [4,15]. They have a peak incidence in the sixth through eighth decades [10]. These tumors most commonly involve the thoracic region (67-84%) and are encountered least commonly in the lumbar region (2-10%) [16,17]. Only a small percentage of meningiomas contain ossification [1,5] and spinal osseous metaplastic meningioma is rare [8,18]. There are some hypotheses as to an etiology of spinal osseous metaplastic meningioma but we do not know yet exactly the cause. The following hypotheses are the contents: (1) psammoma bodies assemble and form bone [1,5]; (2) meningioma cells turn into osteoblasts [7]; and (3) cells from the neural crest differentiate into osteoblasts [6].
It’s not easy to diagnose because of its rareness and surgery for osseous metaplastic meningioma has several difficult aspects. Adhesion of the tumor to the surrounding tissues complicates its dissection and affects the surgical outcome. In our patient, the ossified hard mass was tightly adherent to the normal cord and several rootlets were inserted into the tumor. So, the mass excision was very difficult from the normal cord.
Different long-term outcomes after ossified meningioma surgery have been reported. Roux et al. [15] reported 3 ossified meningiomas out of 54 spinal meningiomas and total resection in 2 cases and a subtotal resection in one case, requiring a second surgery and radiation therapy. After surgery, prognosis was good. Three of the 4 tumors reported by Levy et al. [12] as calcified had a disastrous functional outcome. Total excision of such calcified tumors should not be systematic because of a possible poor neurologic outcome if it has a very close relationship with the spinal cord itself.
Some authors believe that a resection of the dura is necessary to reduce the likelihood of recurrence. However, Klekamp and Samii [9] reported that the recurrence rate in the subset of tumors was independent of management of the dural attachment (resection and duraplasty/coagulation). We coagulated the dura surrounding the tumor.
Ohta et al. [13] reported that positive reaction of MIB antibody to Ki 67 represents the prognosis of meningioma. A positive reaction to Ki 67 in meningiomas demonstrated recurrence. In our case, the Ki 67 antibody was negative staining. The preoperative computed tomography (CT) scan to confirm the ossification and calcification of the tumor seems necessary to evaluate surgical difficulties. Through MRI, CT, and pathology findings, the patient’s postoperative course, they helped us to make a conclusive diagnosis of meningioma.
In conclusion, the patient presented several notable features including (1) rarity; (2) difficulty in surgery because of its characteristics; and (3) the negativity of the immunohistochemical study using Ki 67 antibody represents a favourable prognosis. In addition, a CT scan can give information of difficulty in surgery before the operation.