INTRODUCTION
Carcinoid or neuroendocrine tumors (NETs) are slow-growing neoplasms originating from neuroendocrine cells6,8,17). Although they are known to mainly occur in specific organ systems the lung, pancreas, or gastrointestinal tract, they all share distinct characteristics8). Despite the slow tumor growth, carcinoid tumors can distantly metastasize to other organs, like the liver, and to neuroendocrine cells that secrete serotonin or prostaglandin which may cause carcinoid syndrome5,17).
Limited reports showed the involvement of carcinoid tumors in the central nervous system (CNS). These reports included cases involving primary and metastasis to the CNS. CNS carcinoid tumors induce mass effects, leading to compressive myeloradiculopathy. However, due to their asymptomatic presentation and incidental diagnosis, CNS carcinoid tumors are difficult to diagnose9).
Recent evidence showed that the overall incidence of carcinoid tumors continuously increased16). There was also a growing number of reports on malignant carcinoid tumors16). Although NETs are relatively benign and CNS involvement is rare, the metastatic tendency of the carcinoid is increasing. Moreover, CNS involvement causes irreversible neurologic deficits which affect a patient’s prognosis and quality of life. Therefore, carcinoid tumors, especially those possibly involving the CNS, should not be overlooked, and the clinical importance of accurate diagnosis is emphasized.
This study presented a case of a primary spinal intradural extramedullary carcinoid tumor accompanied by urinary incontinence. CNS involvement in carcinoid tumors is rarely reported, and its intradural spinal metastasis or primary spinal NET is extremely rare5,9). Moreover, due to the indolent progression of carcinoid tumors, a long period of time is needed to observe the occurrence of clinical symptoms before making the diagnosis 13). Hence, by reviewing cases of primary spinal NET, this study aimed to help establish the differential diagnosis in order to accomplish rapid diagnosis and proper treatment while preventing poor outcomes.
CASE REPORT
1. Clinical Presentation
A 63-year-old woman was hospitalized via outpatient clinics for urinary incontinence for 3 years. No distinct somatic motor or sensory abnormalities were observed, and the deep tendon reflexes were normal. Despite her urinary incontinence, electromyography showed no lumbosacral radiculopathy or peripheral neuropathy in the bilateral back and lower limbs, but the patient complained of urinary incontinence. Cystoscopy performed at the Department of Urology revealed no structural anomalies in the urinary bladder, urethral orifice, ureterovesical junction, or urethra. However, a mass lesion was detected at the L5-S1 level on abdominal computed tomography (CT) (Fig. 1A), and a 2.5-cm-sized intradural extramedullary mass was found at the lumbosacral level on spinal magnetic resonance imaging (MRI). The lesion was isointense on T1- and T2-weighted MRI and well-enhanced on contrast images (Fig. 1B, C).
As the patient continued to complain of urinary incontinence without urinary structural problems, and despite the absence of somatic motor and sensory abnormalities, there was a possibility of visceral symptoms due to compression by the lesion detected on MRI, surgery was performed for decompression of the identified mass.
2. Management and Outcome
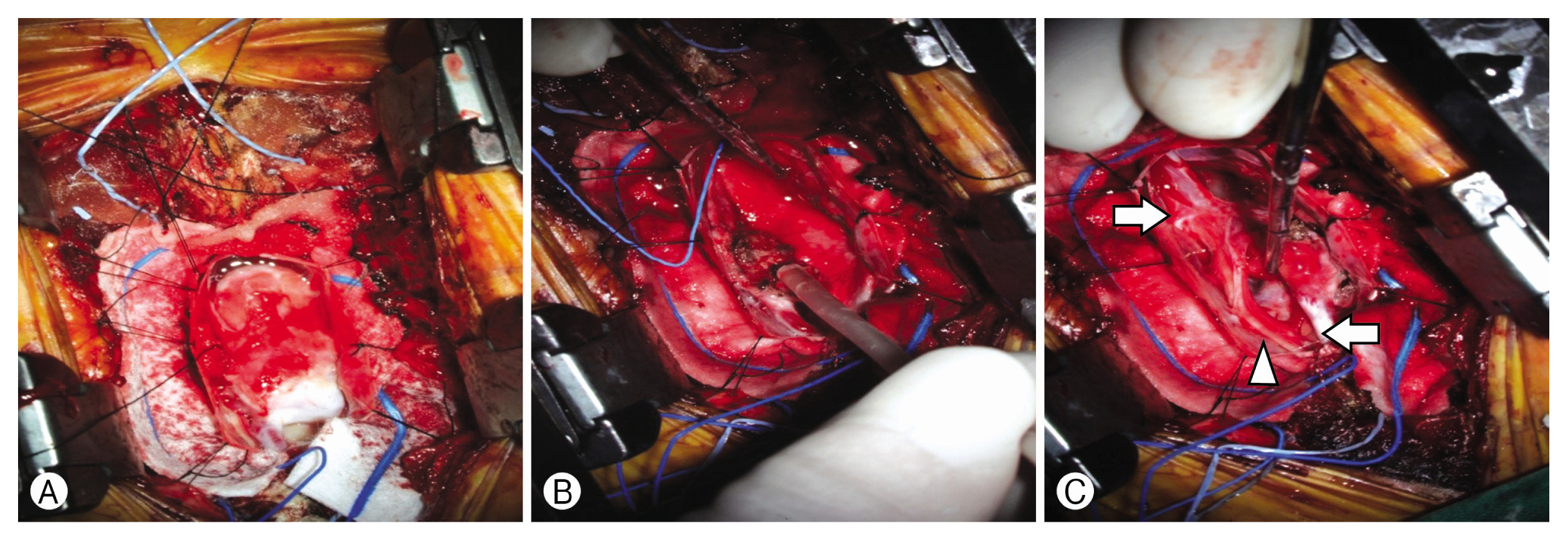
The patient underwent L5-S2 laminectomy to remove the intradural extramedullary mass at the L5-S1 level. The surgery was performed under general anesthesia in a prone position, and abnormal motor or sensory changes were monitored by intraoperative neurophysiological monitoring (IOM; motor evoked potentials and somatosensory evoked potentials). The spinal level was confirmed using a C-arm, and after skin incision from L3 to S3, the lamina was exposed by dissection of the paravertebral muscle. Bilateral laminotomy of the L5 lamina and laminectomy of the S1 and S2 laminae were performed. The removal of the ligament flavum revealed the thecal sac, which was found to be thick, and a linear dural incision was made. The mass size was 3 cm, and dural adhesion was not severe. Before the intradural extramedullary mass was removed, the artery running in proximal and distal was checked with a doppler, and the flow is backflowing from caudal to rostral, so it is decided to preserve without ligation. The doppler observed during surgery confirmed that the arterial flow was located inside the tumor mass in a sigmoid shape, so the tumor was not completely removed and the area around the vessle was preserved. The tumor mass was completely romoved by a peicemeal maneuver using a bipolar coagulator and a cavitron ultrasonic surgical aspirator (Fig. 2). There were no specific changes in IOM.
The patient recovered without complications, and there was an improvement in urinary incontinence. The patient did not complain of urinary incontinence after discharge, and no evidence of recurrence was observed after a 1-year follow-up.
3. Pathologic Findings
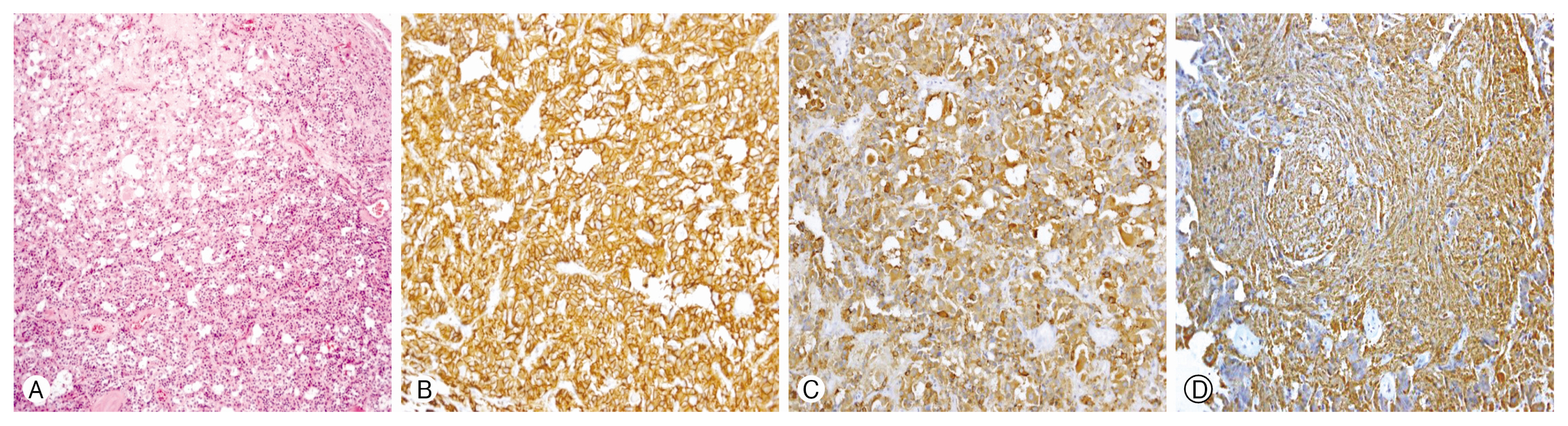
Pathologic examination of the excised mass showed uniform, small, bland tumor cells in solid or glandular patterns on hematoxylin and eosin (H&E) staining (Fig. 3A). Mitotic activity was low (<2/10 high-power fields), and the Ki-67 labeling index was lower than 2%. Immunohistochemistry (IHC) demonstrated the expression of neuroendocrine markers such as CD56, chromogranin, and synaptophysin (Fig. 3B-D). These pathologic findings corresponded to low-grade NETs.
4. Differential Diagnosis Between Primary and Secondary Carcinoid Tumors
After the pathologic diagnosis of low-grade NET, the differential diagnosis between primary and secondary carcinoid tumors was evaluated using various radiologic modalities. Since no specific findings were observed on contrast-enhanced CT in the chest and abdomen performed to confirm the presence of other primary tumors, 18F-fluorodeoxyglucose positron emission tomography (18F-FDG PET) scan and whole-body bone scan (99mTc-Bone Scintigraphy) were performed. On the PET scan, multifocal hypermetabolic lesions were found in the rectosigmoid colon and the left colon, and in the subsequent colonoscopy, the presence of an additional lesion was examined in the whole colon, and the hypermetabolic lesion in the colon was biopsied. There was a 0.3-cm-sized polyp in the left colon, and a tubular adenoma with low-grade dysplasia was confirmed histologically. Therefore, it was confirmed that the hypermetabolic lesion on PET scan was not associated with NET.
DISCUSSION
The term “carcinoid” was first coined by a pathologist, Oberndorfer14) in 1907, which was used to describe distinguishing lesions with a slow-growing nature different from other existing cancers20). In addition, carcinoid tumors are also referred to as NET, which indicates that the tumor cells have both neural and endocrine properties. Neural properties are characterized by the existence of dense-core granules found in serotonergic neurons, and the endocrine property represents the characteristics of producing and secreting these monoamines15).
Since 1963, when NETs were first classified based on the embryonic site of origin, there have been many attempts to classify NETs6,8,10,15). In 2015, the National Comprehensive Cancer Network classified and graded carcinoid tumors and recommended the evaluation of tumor differentiation using a mitotic rate and Ki-67, which is still used today15).
Carcinoid tumors are often diagnosed incidentally. Their incidence accounts for 0.28% to 0.8% per 100,000 people per year, and according to recent evidence, the overall incidence of carcinoid tumors is steadily increasing3,13). Although carcinoid tumors were initially referred to as benign tumors, their propensity to metastasize has recently been discussed13). Recent reports have demonstrated the characteristics of NETs as a wide range of neoplasms, which shows the varied disease course of NET that can be rapidly progressing from a benign disease9). Hence, the treatment and prognosis of carcinoid tumors depend on the location of the primary tumor and the extent of metastasis at the time of diagnosis3).
NETs sometimes show aggressive course or metastasis tendency, and metastatic progression is reported to occur in 22% of cases of malignant NETs, among which the involvement of the CNS is extremely rare, accounting for <2% of the metastases 5,13).
Although the metastatic involvement of carcinoid tumors in the CNS is known to be rare, even rarer cases are primary CNS NETs. This primary CNS NET is so rare that, from 2001 to 2020, only 9 cases have been reported, including the current case (Table 1).
In these 9 cases, the patient age ranged from 30 to 60, with a mean age of 52 years. The male-to-female ratio was almost equal, with a slight male predominance (M:F=5:4).
The tumor location was at the lower cervical and upper thoracic levels in 1 case, and the other 8 cases were found at the lumbosacral level. However, every primary spinal NET in these cases was an intradural extramedullary lesion involving the filum terminale.
The diagnosis of NET was made by postoperative histopathologic examination based on the histological findings with IHC and others, by which the positivity for synaptophysin and chromogranin A was identified in almost all cases.
For the differential diagnosis between primary or secondary NETs, various radiologic modalities and diagnostic tools, including endoscopy, contrast-enhanced CT (chest and abdomen), MRI, 18F-FDG PET, octreotide scan (somatostatin receptor scintigraphy), and 99mTc-Bone scintigraphy were used. In our institution, 18F-FDG PET was performed additionally, and a hyper-metabolic lesion was found in the colon, followed by a biopsy using colonoscopy. The result was a tubular adenoma, which was not histologically consistent with NET, and accordingly, the NET was confirmed to be primary.
In all cases, the first choice of treatment was surgical resection, and grossly total resection was performed if possible. In cases where subtotal resection was performed, or a remnant was found in a postoperative imaging work-up, adjuvant radiotherapy was conducted. It is known that radiation therapy, not applicable as curative therapy, has merits for patients with spinal carcinoid tumor by improving their condition2). Although there is little evidence for the use of radiotherapy or chemotherapy in the treatment of primary spinal carcinoid tumors, postoperative radiotherapy has been reported to prevent tumor recurrence and to reduce the range of the residual lesion10). However, due to the unclear role of chemotherapy and the rarity of primary spinal carcinoid tumors, evidence is still limited to recommend adjuvant therapy5,9).
In summary, in 9 cases including the current case, the following characteristics were observed in the diagnosis of primary carcinoid CNS (spinal) tumor.
First, the location of the primary CNS carcinoid tumor is distinct. Spinal metastatic CNS NET occurs mainly through hematogenous dissemination and usually manifests as epidural in location. In addition, intradural extramedullary location is rare, all of which can be distinguished from primary CNS NET13). Consistent with all 8 previous cases, which were an intradural extramedullary lesion involving the filum terminale, the current case was also intradural extramedullary in location, which corresponds to the feature of primary CNS spinal carcinoid tumors.
Second, IHC plays an important role in histological diagnosis. Along with the established classification and grading of carcinoid tumors using a mitotic rate and Ki-67, specific markers of IHC can help in the diagnosis7). Positive expression of neuroendocrine markers such as CD56, chromogranin, and synaptophysin can be a tool to confirm the diagnosis of NET, and negative expression of markers for primary pulmonary or renal tumors, such as CD10, TTF-1, and RCC or those for meningiomas or schwannomas, such as EMA and S-100, can help to rule out secondary carcinoid tumors4,8,10,18,19). In the 8 cases, negative findings for other IHC markers were reported except in 2 cases, and the current case also confirmed the negativity of various markers.
Third, before the definite diagnosis of a primary carcinoid tumor, it is important to perform a differential diagnosis of metastatic CNS carcinoid tumors by investigating the presence of NETs in other areas of the body. Various radiologic modalities are used for this purpose. For the diagnosis of NET, In-111 octreotide scan using a somatostatin analog as a tracer as well as contrast-enhanced chest and abdominal CT, MRI, 99mTc-Bone scintigraphy, endoscopy, and 18F-FDG PET have been performed1,11). Although octreotide scan is generally known to be the primary diagnostic tool, somatostatin receptor scintigraphy has a tendency to show false-negative results in undifferentiated or poorly differentiated carcinoids, in which 18F-FDG PET could be useful as in general malignant tumors12,15).
CONCLUSION
Carcinoid tumors (NET) are rare, but the overall incidence of carcinoid tumors is steadily increasing, and it is now considered an increase in malignant tumors6,8,10,17). The CNS involvement is reported more rarely, and most cases of CNS NETs are metastases to CNS systems from other primary origins5,9).
Recently, 9 cases of primary CNS carcinoid tumors, especially spinal NETs, have been reported. In every reported case, the diagnosis was confirmed by postoperative histological examination, and positive and negative findings in IHC using various biological markers in addition to the evidence of its primary origin through various radiological modalities could be helpful.