INTRODUCTION
Schwannomas are the most common peripheral nerve sheath tumors8), originating from well-differentiated Schwann cells of the neural sheath of motor and sensory nerves21). They are often solitary, circumscribed and encapsulated by epineurium and grow eccentrically, displacing the nerve root8,22). A malignant potential has been reported in less than 5% of cases and the reported incidence of benign schwannoma is estimated to occur in 6 patients per 100,00024). Sporadic schwannomas have no predilection toward sex or race, and often affect patients aged 20 to 50 years20). They may occur anywhere in all cranial and peripheral nerves except the optic and olfactory nerves.
Schwannomas of the sciatic nerve, which is the largest nerve of the human body, are uncommon (with a frequency of <1% of all schwannomas)20). They could often raise confusion with other known causes of sciatica, such as lumbar degenerative diseases and its diagnosis might be challenging, leading to a late correct diagnosis in many cases24).
Here, we present the case of sciatic nerve schwannoma in a middle-aged female subject who presented with the walking difficulty associated with persistent sciatica.
CASE REPORT
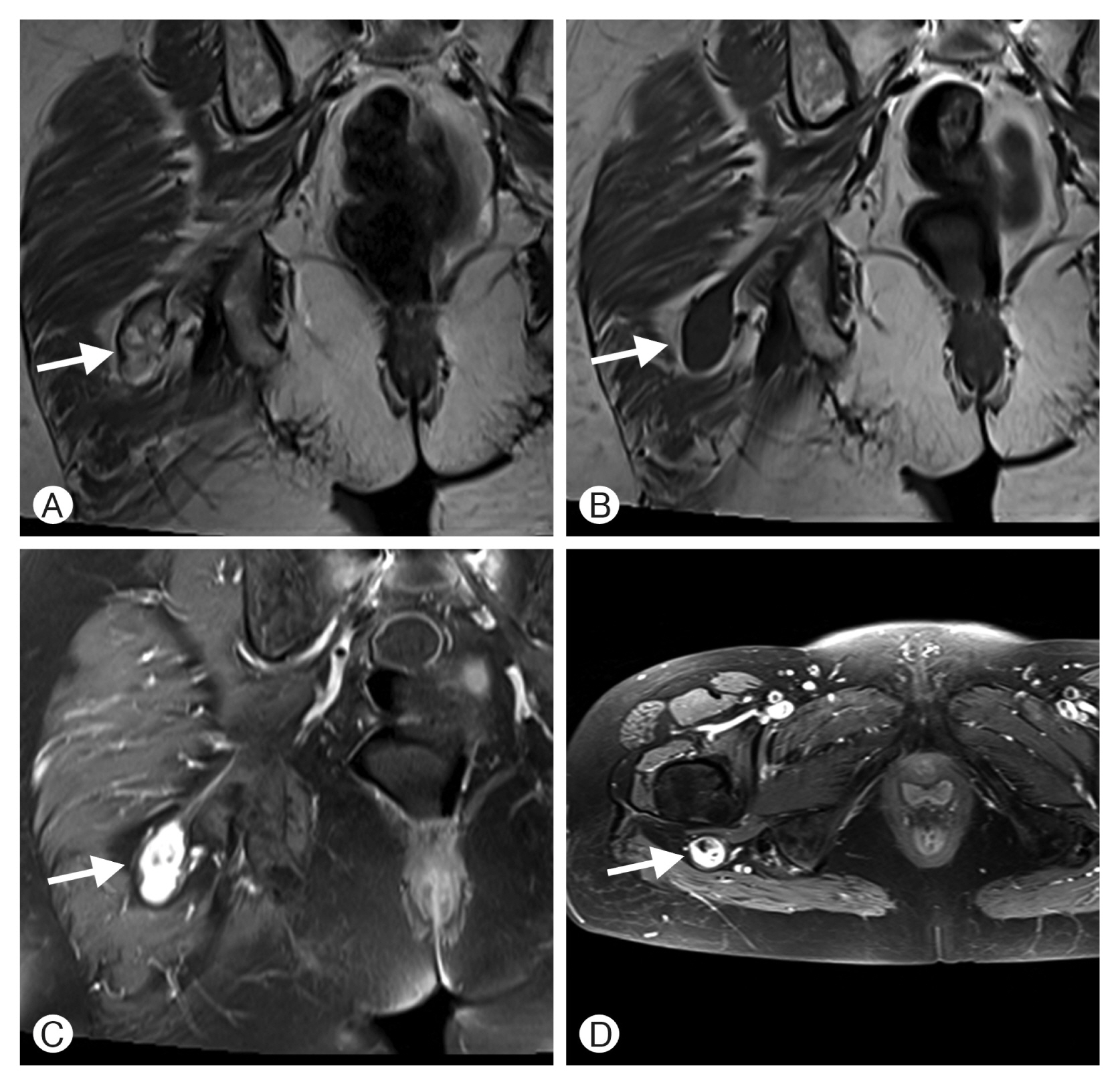
A 63-year-old female presented with pain in the right leg (visual analog scale [VAS] 8/10), which presented for the past 5 years and progressively worsened over time. The pain started in the right buttock area and it radiated down to the lateral thigh, the back of the leg and the dorsum of the foot. The symptom was also present at rest and woke the patient up from sleep during the night. A lumbosacral magnetic resonance image (MRI) scan demonstrated postoperative laminectomy at L4-5 level (Fig. 1A, 1B), which she already has been performed under the diagnosis of spinal stenosis 4 years ago. However, lumbar decompressive surgery failed to relieve her symptom and during the intervening time before the admission, the patient underwent several times of selective nerve root blocks. Following the nerve root block, the relieving effect lasted only for several hr, less than a day at best.
The patient had a history of pulmonary thromboembolism and had been medicated antiplatelet agent for a year. It gave rise to a suspicion of peripheral arterial disease of the legs however, both ultrasound scanning and computed tomography (CT) angiogram showed negative results. Besides, electromyography (EMG) and nerve conduction test documented negative findings and were not contributory. Multiple opinions were sought. The sciatica was thought to originate from compression by piriformis muscle and the patient was sent to the pain clinic to get piriformis block. Ultrasound scanning was performed to localize the piriformis muscle and it revealed a hypoechoic mass in the gluteal region. Subsequent MRI of the gluteal and thigh region was then performed, and it finally found a rounded and heterogeneously enhanced tumor (2.5 x 2 x 3.7 cm diameter; Fig. 2). It went along the course of the right sciatic nerve, posteriorly to the greater trochanter of the right femur. The physical and neurological examination revealed significant pain related to compression of the posterior proximal region of the thigh although the mass was hardly palpable. There was no remarkable motor deficit but hyporeflexia of the right ankle reflex was noted.
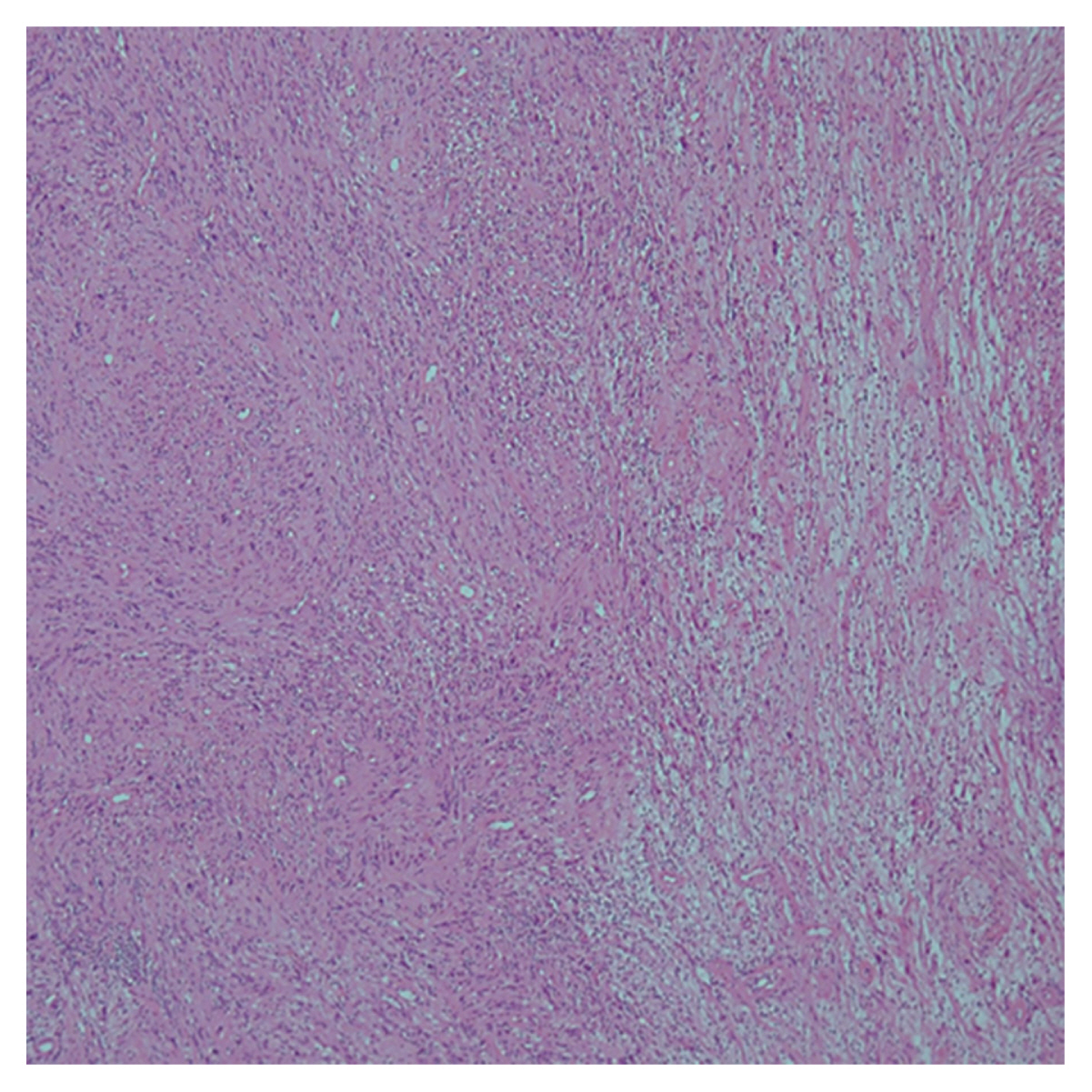
The patient was operated on with microsurgical enucleation with neurophysiological monitoring. The patient was positioned in a left lateral decubitus position and a linear skin incision was made over the greater trochanter. Following the dissection of the iliotibial band and tensor fascia latae, a well-circumscribed, firm, the encapsulated tumor was found originating from the sciatic nerve sheath (Fig. 3A). The nerve sheath was dissected, and the tumor was enucleated from the nerve fascicles, which were incorporated within the capsule. Complete tumor resection was achieved with the preservation of the functioning fascicles (Fig. 3B). Histological analysis of the tumor confirmed the diagnosis of a sciatic nerve schwannoma, owing to the presence of high and low cellular regions called Antoni A and B areas, respectively (Fig. 4).
The postoperative course was uneventful. Prompt and significant decrease of pain (VAS 1/10) and paresthesia were noted allowing rapid mobilization. There was no complication or any newly developed neurological deficits.
DISCUSSION
Sciatica is generally referred to pain radiating the leg, usually below the knee and into the foot and toes. It tends to approximate the dermatomal distribution of the affected nerve root, mostly L5, S1 and often presents paresthesia and numbness in a similar distribution. It may accompany by neurologic deficits such as muscle weakness and reflex change. As with lower back pain is terminology for a symptom rather than a specific diagnosis and typical pathologies included lumbar disc herniation, lumbar canal, or foraminal stenosis. Generally, the main cause of the pain is believed to be attributable to the inflammatory changes resulting from irritation or compression of the affected nerve root by its surrounding structures12). Another potential diagnosis of sciatica includes spinal tumors, spinal epidural abscess/hematoma, myositis ossificans of biceps muscle, facet syndrome, lumbar instability, sacroiliitis, sciatic neuritis, and peripheral nerve sheath tumors10).
The neoplastic involvement of the sciatic nerve has occurred in soft tissue malignancies, sarcoidosis, lymphoma, infiltrating intermuscular lipomas and fibrolipomatous hamartoma6). Filler et al.7) investigated the origin of non-discogenic sciatica in 239 patients and reported that sciatic nerve tumors represented only 1.7% of all cases. Moreover, the incidence of sciatic nerve schwannoma is quite rare that 6 cases per million were reported to occur19,25). Given the fact that schwannomas have a long subclinical course and pain from sciatic nerve schwannoma may simulated sciatica caused by a lumbar disc herniation or spinal stenosis, their clinical presentation is misleading and could lead to late diagnosis (Table 1). The patient in the current case had suffered from sciatic pain for 5 years and was performed lumbar decompressive surgery under the diagnosis of spinal stenosis however, sciatica persisted until the removal of the sciatic schwannoma.
A clinical history of a long-lasting radicular pain being unresponsive to pain medication and rest should raise a suspicion of non-discogenic sciatica and a peripheral nerve lesion. Especially, patients with a negative straight leg raise test coupled with lumbar MRI presenting no sign of disc herniation or stenosis should be performed ultrasonography or hip MRI. Ultrasonography is useful in detecting superficial lesions. Schwannoma usually is detected as a hypoechoic solid and well-centered mass being located eccentrically on the affected nerve with preservation of its fibrillary aspect19). MRI is the gold standard diagnostic tool for peripheral nerve sheath tumors. The mass is well circumscribed and shows an isointense signal on T1-weighted image (WI), with a peripheral hyperintensity and a homogeneous hypointense center on T2-WI. This characteristic MRI finding called a “target sign” could differentiate the schwannoma with neurofibroma, which is usually heterogenous on both T1- and T2-WI23). Cystic degeneration may be noted, if the tumor is subjected to pressure of injury and it can be appeared heterogeneous following gadolinium enhancement whereas schwannoma is usually enhanced homogeneously26).
The physical examination may reveal the palpable lump along the course of the sciatic nerve, which may have tenderness with a positive Tinel’s sign. The neurologic examination often demonstrates the absence of motor and sensory deficit unless the mass is very large. This is due to the lack of an infiltrative nature of the tumor and it also explains the frequent normal findings of EMG examination26). Neural tumors associated with neurologic deficit should always raise the possibility of malignancy9).
To expose the sciatic nerve at the sciatic notch or the gluteal region, a transgluteal or a subgluteal approach is usually employed. With the subgluteal approach, prolonged discomfort from retraction of the soft tissue and the gluteal muscle may ensue. The transgluteal approach could provide a wider surgical corridor up to the sciatic notch however, great care should be taken to muscle arteries, which if not adequately cauterized may retract within the pelvis leading to profound bleeding24).
Surgical excision is the treatment of choice for peripheral nerve schwannomas. Usually, nerve fascicles stretch over the tumor capsule and extracapsular microscopic resection along with gentle dissection of fascicles from the tumor capsule would allow complete enucleation with preservation of the nerve continuity. All intact fascicles should be identified by using intraoperative stimulation which could determine the fascicles that are not functioning, and their transection does not usually end up with additional deficits. The nerve sheath is opened after a wide exposure and isolated from all adjacent neurovascular structures. The entering and exiting fascicles are cautiously identified and divided and the tumor is resected. In large tumors, it firstly necessitates an intratumor debulking and whenever functional fascicles are not possible to dissect from the capsule, it could be a better move to leave a residual mass than provoke neurologic deficit14). However, it was reported that the majority of peripheral nerve schwannomas could be removed totally without significant neurologic deficits13).
Schwannomas are composed of 2 histological patterns: Antoni A and Antoni B. Antoni A is organized with highly vascular hyper-cellular areas, which are packed with Verocay bodies. It represents a circular coalescence of elongated nuclei elongated, spindle-shaped cellular nucleus. Antoni B is the area where the cell mass is coarse with loose myxoid stroma13).
CONCLUSION
Although sciatic nerve schwannoma presents a rare occurrence, it is imperative to take into consideration whenever a patient has long-standing sciatic pain without positive findings of lumbar pathologies. MRI imaging of the nerve is mandatory for diagnosing the lesion. It is essential to outline the course of the sciatic nerve and to delineate the boundary of the mass to preserve the nerve fascicles. A standard microsurgical excision under the guidance of neurophysiologic monitoring will bring a successful removal of the tumor and good postoperative results for the patients.