AbstractObjectiveTo confirm the benefits of vertebroplasty (VP) in selected patients with acute vertebral compression fracture (VCF) and analyze whether the study of a weight dynamic plain lateral radiograph would help in making decisions to perform VP. Using retrospective analysis, we aimed to determine the radiological characteristics of patients benefiting from VP.
MethodsData were collected from 54 patients (age, 56-97 years) diagnosed with osteoporosis and compression fractures between December 2013 and January 2018. Each patient was hospitalized with ≥2 weeks of absolute bed rest (ABR) and treated for osteoporosis. Plain lateral supine radiography of the fractured spine was performed prior to diagnosis, and both supine and standing spinal radiographs were taken 1 and 2 weeks after ABR. Patients were categorized into the VP and non-VP groups 2 weeks after ABR.
ResultsAlthough patients with worse pain and functional progress were selected to undergo VP, patients in the VP group presented better outcomes in the fourth week of evaluation than those in the non-VP group. In a retrospective analysis of the radiographic study, changes in the compression rate between supine and standing (weight-bearing) X-rays (ΔCR), showed a statistically significant correlation with the patient’s outcome in the second week.
ConclusionVP was found to be an effective management option for patients with VCF. A weight-bearing radiographic study of VCFs provided valuable information on patient selection for VP. Therefore, patient selection based on subjective surveys and radiological studies to determine the benefits of VP could be a beneficial management strategy.
INTRODUCTIONVertebral compression fracture (VCF) is a common type of fracture of the skeletal system20). Because most of its etiology is osteoporotic, approximately 1.4 million cases are recorded annually, and its prevalence is increasing as the population ages worldwide13).
VCFs can result in severe pain, spinal misalignment, kyphosis, increased days of bed rest, decreased pulmonary function, inability to perform activities of daily living, depression, and marked reduction in the quality of life and are associated with morbidity and even mortality in patients with osteoporosis24). Thus, adequate prevention and management are essential for reducing these burdens30).
The normal evolution of spinal VCF is reported to be spontaneous consolidation, with progressive decrease in pain during a 6 to 8-week period21). The current guidelines for standard treatment suggest conservative medical care with rest, analgesia, and external bracing22). In addition, with advancements in medical options, including pharmaceuticals, such as teriparatide, romosozumab, and denosumab, conservative osteoporosis treatment has become more effective17). However, conservative care can also exacerbate bone demineralization, and patients may have to tolerate the adverse effects of anti-inflammatory drugs and analgesics28). Furthermore, patients can become dissatisfied with prolonged bed rest because it may cause pneumonia, urinary infection, bedsores, and deep venous thrombosis26). Vertebroplasty (VP) or open surgery, which is performed to prevent abnormal motion, restores vertebral height and corrects kyphosis and may be considered in patients refractory to medical therapy1,9).
VP, in particular, was first developed in France by Galibert et al.11) in 1987. It is an internal fixation method that utilizes medical cement injections, thereby uniting the fragments and preventing fracture fragment motion1,11). This fixation principle in acute fracture pain management has also been well accepted in the management of other skeletal fractures18). However, with the publication of the INVEST study by Kallmes et al.15), the Australian trial by Buchbinder et al.4), and the VERTOS IV trial10), the merits of VP are under debate. Nevertheless, as a relatively non-invasive, low-risk procedure, which is believed to provide immediate and durable pain relief and improve function in patients with painful VCFs, VP is the main method in the management of osteoporotic and malignant vertebral fractures5,6,8).
Selecting a patient who is refractory to conservative care is key to maximizing the potential benefit of VP3). Although the current clinical guideline recommendations have generally been inconsistent and based on clinical and subjective parameters, there is a limit to determining the viability of VP based on clinical status alone22,25). Therefore, our study aimed to determine whether radiological studies provide information on the structural instability of the lesion and contribute to patient selection for VP.
MATERIALS AND METHODSThis retrospective cohort study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the ethics committee prior to study initiation (IRB No. DMC 2019-02-001).
1. Data CollectionThe study was based on data collected from December 2013 to January 2018, comprising 129 patients hospitalized in the emergency room(ER) with acute back pain for <1 week. Among the 129 patients, 54 were finally included in the analysis based on the inclusion and exclusion criteria, and their data were collected from medical charts, radiological images, and surgeons’ records. The inclusion criteria were as follows: (1) acute VCF on magnetic resonance imaging (MRI); (2) age, 50 to 100 years; and (3) osteoporosis diagnosis with bone mineral density (BMD) (T-score ≤-2.5) on a dual energy X-ray absorptiometry. The exclusion criteria were as follows: (1) potential harm to patients with cardiopulmonary conditions (pneumonia, chronic obstructive pulmonary disease, and chronic heart failure) in the absolute bed rest (ABR) state; (2) threecolumn vertebral fracture; and (3) pathological lumbar spine conditions (tumor or infection).
2. Patient Management Protocol and Assessment ToolAll patients were hospitalized from the ER immediately after an acute VCF diagnosis with newly developed back pain. Hospital care was required for all patients for at least 4 weeks, and 2 weeks of ABR was mandated, even during mealtimes or while using the toilet. Opioid and/or non-steroidal anti-inflammatory drugs were administered to control pain. All patients were injected with teriparatide (20 μg daily, SQ) and treated with calcium and vitamin D supplements for osteoporosis.
Patients eligible for VP were selected after 2 weeks of conservative care with mandated ABR. Specifically, patients who were refractory to conservative care, primarily defined as those with a visual analog scale (VAS) score ≥5 or Oswestry Disability Index (ODI) score ≥30 were selected for VP. Among patients with a VAS score of 5, two patients showed prominent improvement in the ODI score, reporting a >5-point decrease in the recent week. As they have shown progress, they were deemed responsive and continued to receive conservative care. Regardless of the selection, conservative care under hospitalization continued for another 2 weeks or more without mandatory ABR, totaling 4 weeks of hospital care at least.
All VP procedures were performed percutaneously by a single neurosurgeon using the +transpedicular method. Patients were then categorized into VP and non-VP groups for the retrospective analysis of radiological and clinical status to determine the characteristics of patients refractory to conservative care and the predictive values that the patients eligible for VP would show. In particular, fracture level, age, sex, body mass index, and bone mineral density (BMD) of the two groups were analyzed.
3. Radiological AssessmentOn the day of admission, MRI and BMD evaluations were performed to confirm the diagnosis of VCF. Initially, supine radiographs of the lateral spine view were obtained from each patient. Radiographs of the lateral view of the non-weight-bearing supine and weight-bearing standing positions under rigid thoracolumbosacral orthosis brace support were taken during the first and second week after hospitalization with ABR. Four weeks after the date of hospitalization, only radiographs of the lateral view of the non-weight-bearing supine position were studied (Fig. 1).
Vertebral body stability was evaluated by comparing the compression rate (CR) with the change in vertebral height of the fractured bone and lordotic angle (LA) in the supine and standing lateral radiographs 1 and 2 weeks after admission. Thereafter, CR was used to calculate the estimated percent ratio of compressed lost vertebral body height to the estimated normal anterior height. The calculation formula is as follows (Equation 1; Fig. 2):
Equation 1. Compression rate (%) formulaLA was measured using the Cobb’s angle made by the upper and lower endplates of the fracture body (Fig. 2). ΔCR was defined as the change in the compression value between the supine (non-weight-bearing) and standing (weight-bearing) radiographs. Specifically, ΔCR was calculated by subtracting the CR value in the standing position from the CR value in the supine position (Equation 2).
The loss of vertebral height and bone edema on MRI was confirmed by radiology specialists to determine compression fractures. Measurements of the radiological features were then performed by two neurosurgeons, and the average value was used for analysis.
4. Clinical AssessmentWe used the VAS and ODI scores at the initial assessment and 1, 2, and 4 weeks after admission. VAS scores for back and leg pain were measured separately on a scale of 1 to 10, with 0 representing no pain and 10 representing the most severe pain imaginable. ODI scores are one of the principal condition-specific outcome measures used in spinal disorder management using a 50-point questionnaire, with 0 representing no disability and 50 representing bed-boundedness or exaggeration of symptoms. Clinical scores of VAS and ODI were surveyed by one nurse specializing in neurosurgery; both score surveys were initially investigated 1 and 2 weeks after ABR and 4 weeks after hospitalization. Patients who underwent VP were additionally surveyed for comparison a day after the procedure.
5. Statistical AnalysisAll analyses were performed using the IBM SPSS 22 program (SPSS Inc., Chicago, IL, USA), and differences were considered statistically significant at p-value less than 0.05. We used the Mann-Whitney nonparametric statistical test corresponding to the χ2 test to compare fracture levels, follow-up periods, VAS scores, ODI scores, CR and ΔCR, patient characteristics, and baseline features between the VP and non-VP groups. Simultaneously, the Wilcoxon signed-rank test medians and interquartile ranges were used to evaluate the effect of VP on the VAS score. Statistical correlations between CR, LA, and VAS and ODI scores were then evaluated post hoc using the Spearman rank correlation analysis for nonparametric data analysis.
RESULTS1. Patient Characteristics and Baseline FeaturesOverall, 54 patients (46 men and 8 women) were included in the study. Their ages ranged from 56 to 97 years, with a mean age (± standard deviation) of 77.13 (±9.28) years. VCFs of traumatic origin was present in six cases, whereas VCFs of spontaneous origin were present in the remaining 48 cases. The average T-score measured for BMD in the spine among patients was −3.61±0.75.
Among the 54 included patients, 30 were selected to undergo VP (VP group), and the remaining 24 continued to undergo conservative care (non-VP group). Fig. 3 shows no significant differences in the fracture level of VCF between the two groups (p=0.430). Moreover, there were no significant differences in the statistical analysis of the two groups in terms of fracture level, sex, BMD, or follow-up period; however, patients in the VP group tended to be significantly older than those in the non-VP group (p=0.040; Table 1).
2. Radiological Outcomes During Each Follow-upThe CR and LA of the patients on each period of hospitalization did not show statistically significant changes with time (Table 2, 3).
On initial evaluation, the CR and LA of the VP group were measured to be 23.52%±16.49% and 10.81°±6.34°, respectively. In the non-VP group, the CR was 25.36%±16.88% and LA was 11.18°± 5.12°. No significant differences were found in the CR and LA between the two groups.
At the follow-up after 1 week of ABR, the CR in the supine and standing positions was 24.56%±13.06% and 32.58%±12.79%, respectively, in the VP group. The corresponding CR measurements in the non-VP group were 27.15%±15.66% and 29.62%±14.77%, respectively. The LA in the supine and standing positions were 11.13° ±5.23° and 13.78°±5.74°, respectively, in the VP group and 11.40° ±6.23° and 12.29°±6.14°, respectively, in the non-VP group. Similar to the initial results, no significant differences were observed in the CR and LA between the two groups during the 1-week follow-up period.
At the follow-up after 2 weeks of ABR, the CR in the supine and standing positions was 26.40%±14.55% and 37.22%±14.33%, respectively, in the VP group and 30.61%±17.32% and 33.65%±17.17%, respectively, in the non-VP group. The LA in the supine and standing positions was 11.87°±5.76° and 13.08°±6.90°, respectively, in the VP group and 12.59°±11.87° and 13.39°±6.39°, respectively, in the non-VP group. Similar to the previous results, no significant differences were observed between the two groups regarding CR and LA during the 2-week follow-up period At the follow-up after 4 weeks of ABR, the ΔCR values of the non-VP and VP groups were 2.0%±6.12% and 8.2%±5.86%, respectively, showing statistical significance (p=0.010; Fig. 4, 5). At the 2-week follow-up, the ΔCR values of the non-VP and VP groups were 3.04%±4.58% and 9.98%±5.61%, respectively (p=0.010).
The CR at each period showed a statistical correlation with the corresponding LA at the respective follow-up points (p<0.001).
3. Clinical OutcomesThere were no statistically significant differences between the VP and non-VP groups at the initial evaluation and the 1-week follow-up. On initial evaluation, the VAS and ODI scores in the VP group were 7.63±0.85 and 34.03±0.85, respectively, whereas those in the non-VP group were 7.66±0.81 and 33.91±2.20, respectively.
At the 1-week follow-up, the VAS and ODI scores in the VP group were 5.06±0.63 and 33.46±1.99, respectively, and those in the non-VP group were 5.04±0.69 and 33.16±2.21, respectively.
At the 2-week follow-up, the VAS and ODI scores in the VP group were 4.96±0.66 and 31.23±1.79, respectively, and those in the non-VP group were 3.95±0.55 and 26.12±2.07 (p=0.000 and p=0.000), respectively.
A day after VP, the VP group showed a statistically significant improvement in the mean VAS score from 4.96±0.66 to 2.2±0.76 (p=0.000).
At the 4-week follow-up, the VAS and ODI scores in the VP group were 1.8±0.61 and 7.43±2.14, respectively, whereas those in the non-VP group were 2.83±1.09 and 16.70±1.54 (p=0.000 and p=0.000), respectively. Both the groups showed gradual improvement, with the VP group showing a significant turnover and faster recovery (Table 4, Fig. 6).
4. Correlations between Radiological and Clinical OutcomesInitial and 1-week follow-up evaluations of the VAS and ODI scores showed no correlation with radiological features.
However, there was a statistically significant correlation between the VAS and ODI scores surveyed in the second week and the ΔCR values at the 1 and 2 weeks. Specifically, the ΔCR value at 1 week of ABR was correlated with the VAS (p=0.008; R=0.168) and ODI (p=0.008; R=0.359) scores during the second week. Similarly, the ΔCR value at 2 weeks of ABR showed a correlation with the VAS (p=0.001; R=0.542) and ODI (p=0.000; R=0.572) scores at the second week.
Moreover, the ΔCR value at 1 week of ABR showed a correlation with the ODI score at the fourth week with the reversed correlation coefficient (p=0.001; R= −0.431), and the ΔCR value at the second week showed a correlation with the VAS (p=0.009; R= −0.437) and ODI (p=0.029; R= −0.370) at the fourth week.
DISCUSSIONAs the population ages worldwide, the prevalence of VCFs continues to increase22). However, with the rapid pain alleviation provided by chemical/thermal neurolytic effects and fractured vertebrae stabilization, VP has become readily available under local anesthesia, has been deemed relatively safe, and is widely considered a costeffective option2,7,8). Thus, with the increase in the prevalence and benefits of VCF, more patients have been receiving VP for VCF treat ment.
With recent randomized trials showing conflicting results and with the advancing efficacy of conservative care, the benefits of the procedure have been reviewed extensively and repeatedly4,7,15-17,27). Although few randomized trials have failed to demonstrate the benefits of VP, several studies have reported that VP is a valid management option, as long as proper patient selection is assured10,12). Consequently, the current recommendations for VP treatment emphasize sufficiently robust patient selection.
However, the criteria recommended by these guidelines are based on clinical and subjective parameters25). Korea’s national insurance recommends consideration for VP when conservative treatments fail to achieve sufficient pain relief after 2 weeks. Even in-depth reviews from researchers have suggested the criteria for accurate diagnosis, including a shorter pain duration, severe pain, and ununited fracture on MRI19,29). Among these, pain, as the main factor for making decisions to perform VP, depends on the patient’s subjective complaint. As such, quantifiable parameters have a niche over subjective surveys in making decisions to perform invasive procedures.
Therefore, our analysis hypothesized that the characteristics of patients who may benefit from VP reflect not only pain and functional indices but also radiological features of the affected area.
After 2 weeks of conservative care, we treated eligible patients with acute VCF with VP based on pain and functional indices. We then compared the outcome of the VP group to that of the non-VP group (conservative care only), and post hoc analysis was performed on radiographic data collected from the beginning and throughout the period of hospitalization. Although patients with worse pain and functional progress were selected for VP, the symptoms of patients in the VP group showed rapid relief and presented even better outcomes during the 4-week follow-up than those in the non-VP group.
Consistent with the analysis results of a previous study14), our results demonstrated that the calculated CR showed a relationship with the corresponding measured LA. Although a study has shown a relationship between back pain and hyperkyphosis severity23), LA and CR did not show a statistical correlation with the pain and disability indices. According to our results, kyphosis as a radiological feature did not represent a patient’s acute symptom. Furthermore, in VCFs, aside from kyphosis, the motion of fractured particles in the vertebra may indicate the patient’s status. The ΔCR, which is the measure of vertebral compression on weight-bearing, at 1 and 2 weeks after ABR showed statistically significant correlations with the VAS and ODI scores in the second week. This indicated that the fractured vertebra had not been healed to withstand the body weight applied, and macroscopic motion of these fractured segments was observed using dynamic radiography1,29). Furthermore, the VP group being refractory to conservative care could be attributed to these macroscopic and microscopic motions in the fractured vertebra, resulting in larger ΔCR values18). Therefore, cement augmentation in these patients should be beneficial.
In the VP group, the analysis of ΔCR values at 1 and 2 weeks after ABR showed a correlation with patient outcomes at the fourth week using a reversed correlation coefficient. This relationship was considered to be due to the selection of favorable outcomes of VP.
Comparing the radiological features of the two groups, ΔCR of the VP group at the 1- (8.02±5.86%) and 2-week periods (9.98±5.61%) showed a statistically significant increase. The VAS score also showed a correlation with ΔCR at the corresponding periods; however, the differences between the two groups were close to 1 point (VP group, 4.967±0.66; non-VP group, 3.95±0.55). Relying on one point of a subjective survey for making a decision in invasive surgery is incomplete in many aspects; thus, considering quantitative values can strengthen the decision-making process.
Moreover, though patients were categorized into VP and non-VP groups based on the VAS and ODI scores of the 2nd week, no statistical differences of the VAS and ODI scores were shown in data during the initial and even the 1st week of conservative care. In contrast, by retrospective analysis, the ΔCR value of the future VP group showed differences in the 1st week. Since patient selection was made by 2nd week’s VAS and ODI scores, and that these parameters surveyed on 1st week did not predict the 2nd week’s selection, 1st week’s ΔCR showing statistical differences gives ΔCR the potential to be useful as a predictive value in future patient management and selection for VP.
Though we found a statistical difference in the ΔCR value with patients who showed worse clinical outcomes after 2 weeks of conservative care, and these patients gained benefits from VP, our study is limited by its retrospective design. We believe a prospective cohort study with the ΔCR value as a patient selection tool could elucidate the matter. In addition, the study had a relatively small sample size and did not involve a sham procedure to eliminate a placebo. Moreover, although hospitalization care and evaluation for patient outcome were continued for at least 4 weeks, measurement and analysis of CR and ΔCR were only performed for the 1-week and 2-week radiograph images. Although the results were considered to be relatively reliable, as all patients received care under the same and controlled environment with ABR as a principal to eliminate bias, a study with a larger cohort is needed to confirm these results. Furthermore, the varying bone quality of the patients was quantified using BMD only. Further evaluation (such as performing quantitative computed tomography) would help reduce this bias.
CONCLUSIONThe decision to perform VP in patients with VCF is challenging. With conflicting results and indistinctive evidence, patient selection is considered key to VCF management. Although the current clinical guidelines are generally inconsistent, both clinical parameters and radiological features are helpful tools for determining the need for invasive intervention. Thus, radiological features provided by weight-bearing radiographs could benefit patient selection and ensure more favorable outcomes, especially for those with difficulties in responding to the survey.
Fig. 1Imaging timeline of the study. MRI: magnetic resonance imaging; BMD: bone mineral density; VP: vertebroplasty. 
Fig. 2Compression rate (%) calculation. Compression rate is calculated by subtracting the anterior body height of the fractured body (B) from the average anterior height of the upper (A) and lower (C) vertebral bodies and then dividing the difference by the average anterior height of the upper and lower vertebral bodies, which is converted into percentage. Lordotic angle (α°) was measured by the angle made by the upper and lower endplates of the fracture body. 
Fig. 4Radiographic study of a patient in the non-vertebroplasty group. Initial compression rate=12%, first-week supine compression rate=14%, first-week standing compression rate=25%, first-week Δcompression rate=11%, second-week supine compression rate=21%, second-week standing compression rate=21%, second-week Δcompression rate=0%, and fourth-week compression rate=27%. 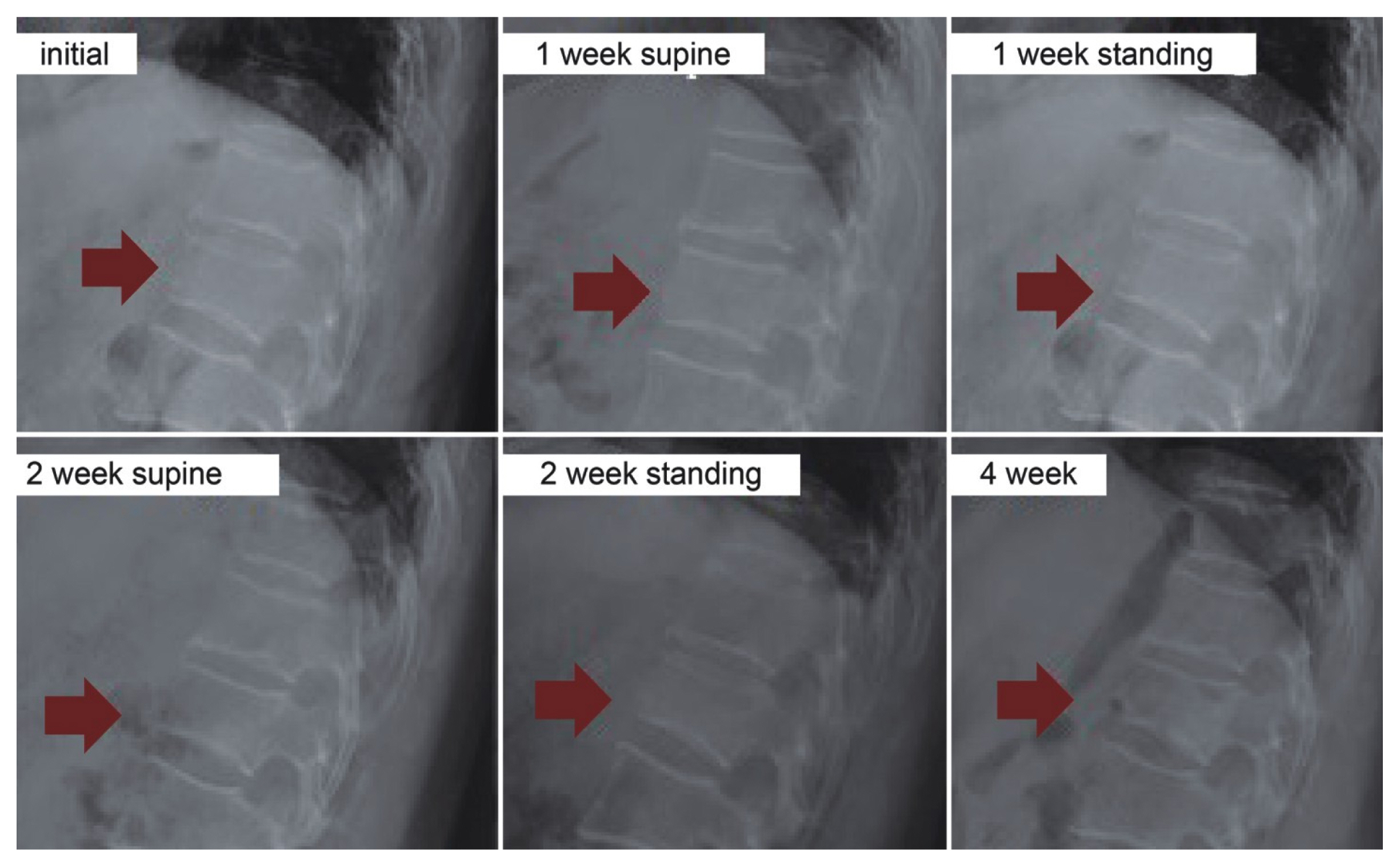
Fig. 5Radiographic study of a patient in the vertebroplasty group patient. Initial compression rate= 38%, first-week supine compression rate=38%, first-week standing compression rate=47%, first-week Δcompression rate=9%, second-week supine compression rate=34%, second-week standing compression rate=56%, second-week Δcompression rate=22%, and fourth-week compression rate 34%. 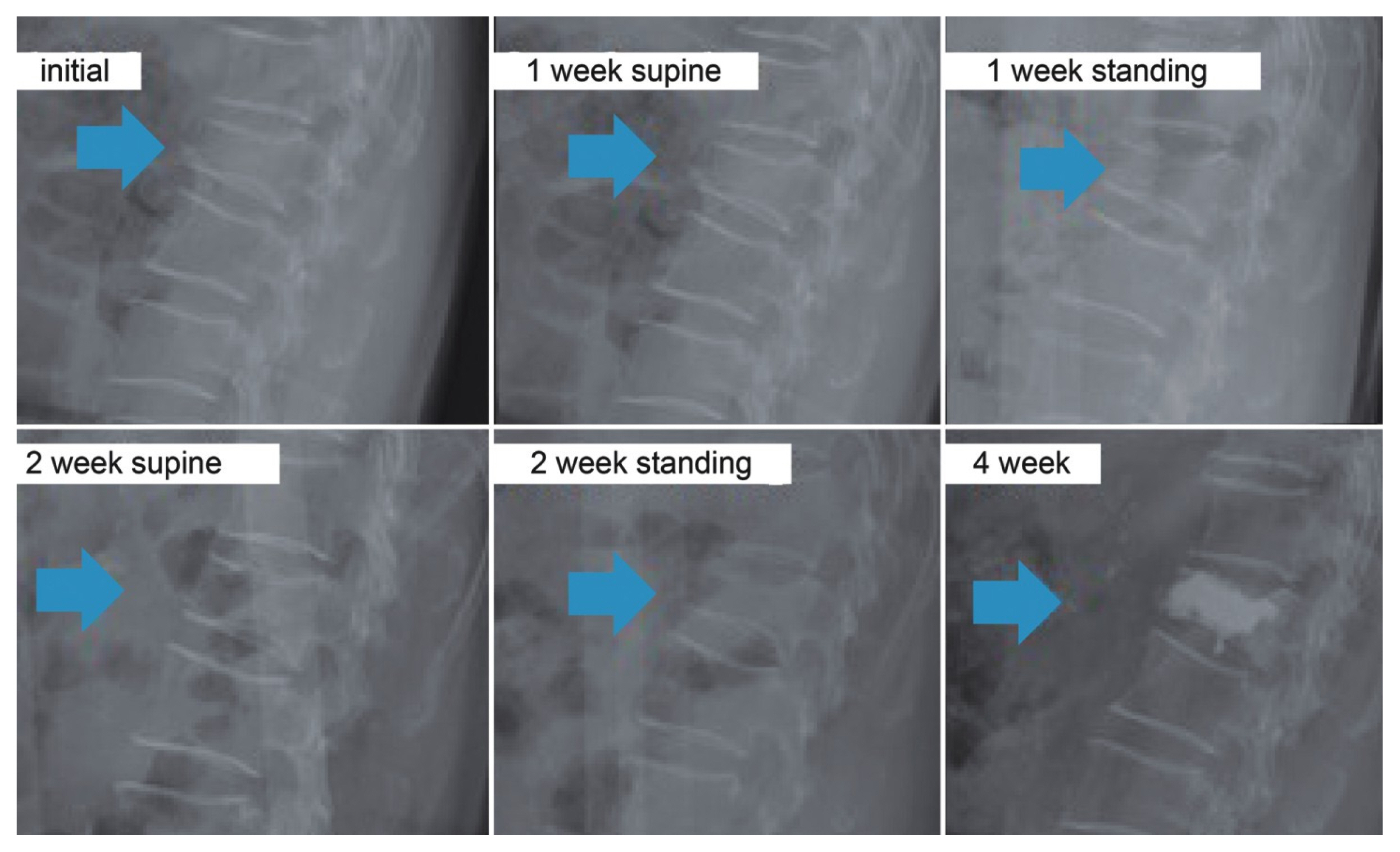
Fig. 6Pain and functional outcomes in the vertebroplasty (VP) and non-VP groups. The VP group showed better outcomes after undergoing VP. VAS: visual analog scale; ODI: Oswestry Disability Index. 
Table 1Patient characteristics in the VP and non-VP groups Table 2CR and change in CR on weight bearing in the VP and non-VP groups Table 3LA in the VP and non-VP groups Table 4Pain and functional outcomes based on the VAS and ODI scores between the VP and non-VP groups
REFERENCES1. Barr JD, Barr MS, Lemley TJ, McCann RM: Percutaneous vertebroplasty for pain relief and spinal stabilization. Spine (Phila Pa 1976) 25:923-928, 2000
2. Baumann C, Fuchs H, Kiwit J, Westphalen K, Hierholzer J: Complications in percutaneous vertebroplasty associated with puncture or cement leakage. Cardiovasc Intervent Radiol 30:161-168, 2007
3. Brodano GB, Amendola L, Martikos K, Bettuzzi C, Boriani L, Gasbarrini A, et al.: Vertebroplasty: benefits are more than risks in selected and evidence-based informed patients. A retrospective study of 59 cases. Eur Spine J 20:1265-1271, 2011
4. Buchbinder R, Osborne RH, Ebeling PR, Wark JD, Mitchell P, Wriedt C, et al.: A randomized trial of vertebroplasty for painful osteoporotic vertebral fractures. N Engl J Med 361:557-568, 2009
5. Clark W, Bird P, Gonski P, Diamond TH, Smerdely P, McNeil HP, et al.: Safety and efficacy of vertebroplasty for acute painful osteoporotic fractures (VAPOUR): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet 388:1408-1416, 2016
6. Diamond TH, Champion B, Clark WA: Management of acute osteoporotic vertebral fractures: a nonrandomized trial comparing percutaneous vertebroplasty with conservative therapy. Am J Med 114:257-265, 2003
7. Dong R, Chen L, Tang T, Gu Y, Luo Z, Shi Q, et al.: Pain reduction following vertebroplasty and kyphoplasty. Int Orthop 37:83-87, 2013
8. Evans AJ, Jensen ME, Kip KE, DeNardo AJ, Lawler GJ, Negin GA, et al.: Vertebral compression fractures: pain reduction and improvement in functional mobility after percutaneous polymethylmethacrylate vertebroplasty retrospective report of 245 cases. Radiology 226:366-372, 2003
9. Firanescu CE, de Vries J, Lodder P, Schoemaker MC, Smeets AJ, Donga E, et al.: Percutaneous vertebroplasty is no risk factor for new vertebral fractures and protects against further height loss (VERTOS IV). Cardiovasc Intervent Radiol 42:991-1000, 2019
10. Firanescu CE, de Vries J, Lodder P, Venmans A, Schoemaker MC, Smeets AJ, et al.: Vertebroplasty versus sham procedure for painful acute osteoporotic vertebral compression fractures (VERTOS IV): randomised sham controlled clinical trial. BMJ 361:k1551, 2018
11. Galibert P, Deramond H, Rosat P, Le Gars D: Preliminary note on the treatment of vertebral angioma by percutaneous acrylic vertebroplasty. Neurochirurgie 33:166-168, 1987
12. Gangi A, Clark WA: Have recent vertebroplasty trials changed the indications for vertebroplasty? Cardiovasc Intervent Radiol 33:677-680, 2010
13. Johnell O, Kanis JA: An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int 17:1726-1733, 2006
14. Kado DM, Browner WS, Palermo L, Nevitt MC, Genant HK, Cummings SR: Vertebral fractures and mortality in older women: a prospective study. Study of Osteoporotic Fractures Research Group. Arch Intern Med 159:1215-1220, 1999
15. Kallmes DF, Comstock BA, Heagerty PJ, Turner JA, Wilson DJ, Diamond TH, et al.: A randomized trial of vertebroplasty for osteoporotic spinal fractures. N Engl J Med 361:569-579, 2009
16. Klazen CA, Verhaar HJ, Lampmann LE, Juttmann JR, Blonk MC, Jansen FH, et al.: VERTOS II: percutaneous vertebroplasty versus conservative therapy in patients with painful osteoporotic vertebral compression fractures; rationale, objectives and design of a multicenter randomized controlled trial. Trials 8:33, 2007
17. Kobayakawa T, Miyazaki A, Saito M, Suzuki T, Takahashi J, Nakamura Y: Denosumab versus romosozumab for postmenopausal osteoporosis treatment. Sci Rep 11:11801, 2021
18. Lewis GS, Mischler D, Wee H, Reid JS, Varga P: Finite element analysis of fracture fixation. Curr Osteoporos Rep 19:403-416, 2021
19. Lim J, Choi SW, Youm JY, Kwon HJ, Kim SH, Koh HS: Posttraumatic delayed vertebral collapse: Kummell’s disease. J Korean Neurosurg Soc 61:1-9, 2018
20. Melton LJ 3rd, Thamer M, Ray NF, Chan JK, Chesnut CH 3rd, Einhorn TA, et al.: Fractures attributable to osteoporosis: report from the National Osteoporosis Foundation. J Bone Miner Res 12:16-23, 1997
21. Pappou IP, Papadopoulos EC, Swanson AN, Cammisa FP Jr, Girardi FP: Osteoporotic vertebral fractures and collapse with intravertebral vacuum sign (Kümmel’s disease). Orthopedics 31:61-66, 2008
22. Parreira PCS, Maher CG, Megale RZ, March L, Ferreira ML: An overview of clinical guidelines for the management of vertebral compression fracture: a systematic review. Spine J 17:1932-1938, 2017
23. Ryan PJ, Blake G, Herd R, Fogelman I: A clinical profile of back pain and disability in patients with spinal osteoporosis. Bone 15:27-30, 1994
24. Silverman SL: The clinical consequences of vertebral compression fracture. Bone 13(Suppl 2):S27-S31, 1992
25. Stallmeyer MJ, Zoarski GH, Obuchowski AM: Optimizing patient selection in percutaneous vertebroplasty. J Vasc Interv Radiol 14:683-696, 2003
26. Suzuki N, Ogikubo O, Hansson T: The course of the acute vertebral body fragility fracture: its effect on pain, disability and quality of life during 12 months. Eur Spine J 17:1380-1390, 2008
27. Uebelhart B, Ferrari S: Romosozumab : a new treatment for severe osteoporosis. Rev Med Suisse 17:784-787, 2021
28. Whedon GD: Disuse osteoporosis: physiological aspects. Calcif Tissue Int 36(Suppl 1):S146-S150, 1984
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||