INTRODUCTION
Meningiomas are the most prevalent primary brain tumors; however, primary extracranial meningioma (PEM) is uncommon5). PEM is an extracranial meningioma not connected to a meningioma in the neuraxis. Therefore, in the diagnosis of PEM, the extension of central meningioma should be excluded. PEMs most commonly occur in the skin, especially the scalp and middle ear, followed by the nasal cavity14). They have also been found in the neck, intra-abdominal pelvis, and orbit. Despite being found in various locations, their morphology and histological characteristics are similar to those of their intracranial counterparts. Although surgical treatment is considered the priority for PEM, radiation therapy may also be considered in the case of a recurrence. The primary goal of surgical treatment is total resection of the tumor5). We have experienced a case of PEM in the scalp with invasion of the skull, and here we describe our unique case.
CASE REPORT
A 74-year-old woman was admitted to our department with a 2-year history of a right frontotemporal lesion on the scalp and right periorbital swelling which rapidly progressed within three months. On physical examination, the mass was hard and fixed without tenderness or redness, and there were no neurological symptoms.
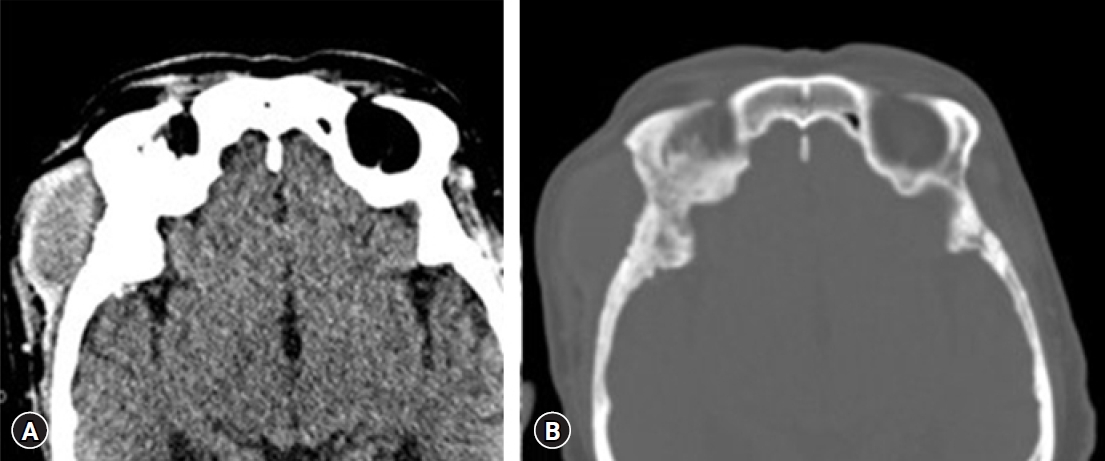
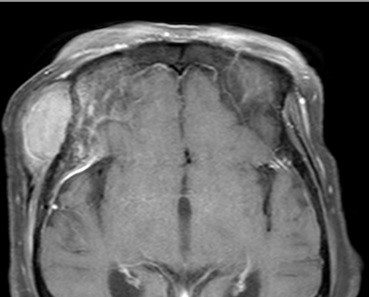
A soft tissue mass of about 4.5 cm in maximum diameter with homogeneous attenuation and mild enhancement in the right infratemporal fossa area was identified (Fig. 1A), and diffuse cortical thickening of the adjacent skull was identified on the computed tomography scan (Fig. 1B). An magnetic resonance imaging revealed a homogeneously enhanced mass involving the right frontotemporal scalp, temporalis muscle, and right frontal and temporal bone. An osteolytic lesion was seen in the sphenoid bone and diffuse dural thickening and enhancement were found just under the osteolytic site (Fig. 2).
A curvilinear scalp incision was made in the right frontotemporal area. Under the scalp flap with the temporalis muscle, a tumor mass was identified, and gross total removal of the scalp tumor was performed. The bone invasion was checked and removed with a 1 cm tumor-free margin. The internal surface of the craniotomy flap was not involved with the tumorous lesions. Also, there was no visual abnormality of dura after craniotomy was performed. It was judged that additional measures such as dura excision would not be necessary. The defect of the skull was covered with artificial bone cement (Fig. 3). The tumor mass was sent to the pathologist for sectioning and was determined to be myosarcoma.
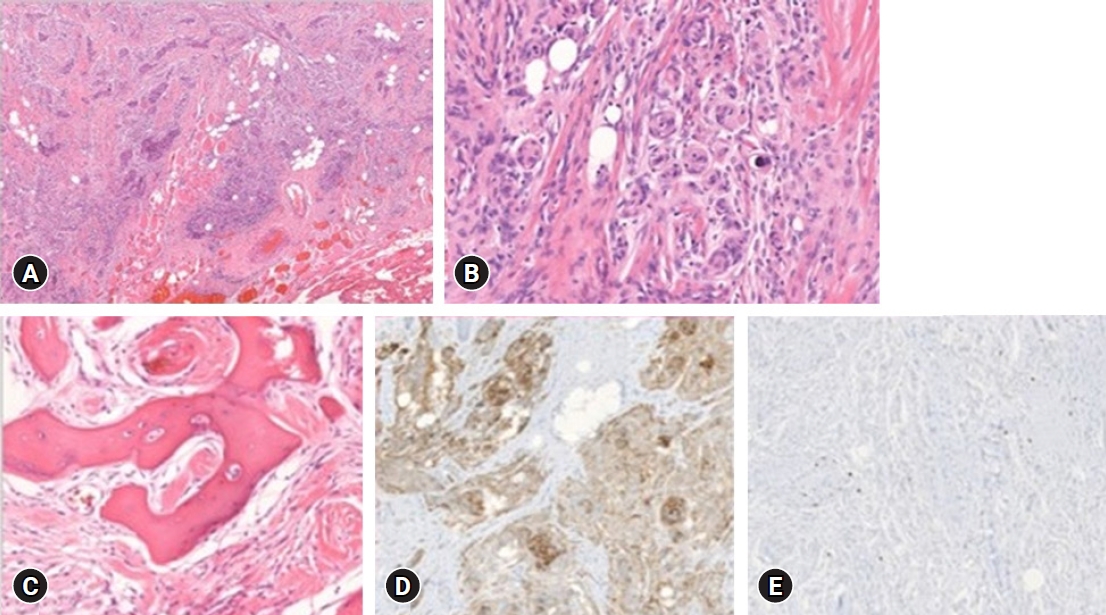
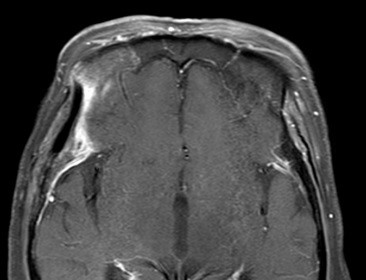
The resected soft tissue mass was 5.0 x 3.6 cm in size, grossly. The pathologist confirmed that the scalp mass was meningothelial meningioma with increased cellularity. A cranium bone biopsy was also done at the craniectomy site, and involvement of meningothelial meningioma was demonstrated. Microscopically, skeletal muscle and fat tissue were infiltrated by the meningioma (Fig. 4A). Typical whorls, syncytial structures, and sparse psammoma bodies with no cytologic atypia and rare mitosis (less than 1 per 10 high power fields) (Fig. 4B) revealed the diagnosis of meningothelial meningioma, World Health Organization (WHO) grade I. The tumor involved the bone marrow of the temporal cranium (Fig. 4C). Epithelial membrane antigen (EMA) immunohistochemical staining revealed diffuse positivity (Fig. 4D). The proliferation index confirmed by the Ki-67 antibody was under 2% (Fig. 4E). There was no sign of tumor recurrence and complications after four years of follow-up. (Fig. 5) This case report was approved by the Institutional Review Board (IRB) of Inje University Busan Paik Hospital (IRB No. 2021-10-020).
DISCUSSION
We have described a woman with a meningioma located extradurally, presenting with a scalp mass in the right frontotemporal area. The tumor was diagnosed initially as a myosarcoma arising from the temporalis muscle because of an osteolytic lesion and diffuse dural thickening. However, as a result of permanent pathology, it was confirmed as WHO grade 1 meningothelial meningioma.
Meningiomas are the most common primary brain tumors that arise from the meninges12). The four most common locations are the parasagittal regions, the tuberculum sellae, the convexity, and the sphenoid ridge4). They originate from arachnoid cap cells which make up the outer membrane of the arachnoid mater13). In a recent study, it was found that they constitute approximately 30% of all intracranial tumors in adults. Their incidence increases with age, where they are relatively rare in children and adolescents (0.4%-4.6%)9). However, PEMs have rarely been reported and are frequently misdiagnosed due to their infrequent occurrence14). In the head and neck area, PEM has been found in the facial bone, orbit, middle ear cavity, nasal cavity, temporal bone, and paranasal sinuses8). The incidence of meningioma in the head and neck area was in the order of the scalp, temporal bone and ear, nasal and paranasal area, and orbital structure15). Epithelial tumors, melanoma, olfactory neuroblastoma, angiofibroma, paraganglioma, and ossifying fibroma must be distinguished from PEMs2).
Hoye et al.7) proposed the following classification system for extracranial meningiomas: Type A, extracranial extensions of a meningioma with an intracranial origin (secondary); Type B, extracranial extensions of a meningioma arising in a neural foramen (primary); Type C, ectopic without any connection either to a foramen of a cranial nerve or to intracranial structures (primary); and Type D, extracranial metastasis from an intracranial meningioma (secondary) (Table 1). Most extracranial meningiomas are secondary to a primary intracranial tumor. Primary meningioma without a connection to an intracranial component or a cranial nerve is extremely rare (0.9%), similar to the present case1).
Various theories explain the rise of extradural meningiomas. Firstly, abnormal migration of arachnoid cells is a possible option, whereby migration could occur during embryologic development or birth, during fusion of the suture line, or at the time of head trauma6). Secondly, meningiomas may arise from the dura penetration points of cranial nerves, which would explain the development of paranasal sinus and orbit extradural meningiomas. Arachnoid cells form clusters surrounding the dura exiting point of cranial nerves 3, 7, and 9 to 1210). Lastly, because meningiomas are mesenchymal tumors, multipotential mesenchymal cells which locate extradural might differentiate into extradural meningiomas3,10).
For the diagnosis of PEM, immunohistochemical investigations are necessary. Most extracranial meningiomas have a varied immunohistochemical profile for vimentin, EMA, cytokeratin, S-100, desmoplakin, and desmin. EMA and vimentin are immunopositive in more than 95% of all meningiomas11).
In our case, the patient underwent total tumor resection, and the bone flap was also widely resected. Because of an osteolytic lesion and involvement of the temporalis muscle, malignancy such as osteogenic sarcoma or myosarcoma was suspected before the operation. Meningothelial meningioma, WHO grade I, was diagnosed, and without adjuvant therapy, there has been no recurrence of the tumor in four years of follow-up. According to the classification of Hoye et al.7), Type C, ectopic without any connection either to a foramen of a cranial nerve or to intracranial structures, is the type of this case. Because there is no meningioma in the intracranium, it can be considered a primary type first, and there is no connection with the above structures.
Since recurrence of meningioma, WHO grade I is rare following adequate surgical resection, this case supports the notion that total surgical excision without adjuvant therapy is the treatment of choice for primary EM.
CONCLUSION
PEM is a very rare disease that is easy to mistake for other tumors. In patients with slow growth of tumors in the head and neck, PEM needs to be considered. Meningioma, WHO grade I rarely recurs after proper surgical resection, so complete surgical resection without adjuvant therapy is a good treatment option for PEM.