Contrast-Induced Encephalopathy after Coil Embolization: A Case Report and Review of Literature
Article information
Abstract
Contrast-induced encephalopathy (CIE) is a rare and unexpected complication after coil embolization to treat cerebral aneurysm. A 66-year-old woman underwent secondary coil embolization with iodixanol for recanalization of the left middle cerebral artery aneurysm. Preoperative computed tomography revealed encephalomalacia in the left frontal and temporal lobe caused by previous aneurysmal rupture. Two hr after the procedure, the patient showed status epilepticus and right hemiplegia. Fluid-attenuated inversion recovery imaging revealed extensive involvement of CIE in the left hemisphere. CIE-related symptoms gradually resolved with conservative treatment including antiepileptic drugs, mannitol, and methylprednisolone. Two-month follow-up magnetic resonance imaging revealed that the edema markedly improved in the parietal area but remained in the frontal area, and the patient completely recovered without any sequelae. Even iodixanol, an iso-osmolar contrast medium, could lead to CIE manifesting as a serious condition such as status epilepticus. Endovascular neurosurgeons should always be aware of CIE, especially when encephalomalacia is present.
INTRODUCTION
Contrast-induced encephalopathy (CIE) is an unexpected neurological complication after endovascular procedures with contrast media2,3,12). This complication has a rare incidence that broadly ranges from 0.3% to 1% but might increase to 4% when a hyperosmolar contrast is used13). Its clinical presentations are mainly characterized by transient cortical blindness, headache, memory loss, hemiparesis, aphasia, loss of coordination, confusion, seizures, and coma15). A convincing theory of CIE is that hyperosmolar contrast agents remove fluid from the endothelial cells, causing them to disrupt the tight junctions of the blood-brain barrier17).
Iodixanol, which is the only iso-osmolar contrast medium, has a more favorable neurotoxicity profile than monomeric, nonionic contrast media1,16). The association between CIE and iodixanol contrast agents has rarely been reported3,12). Here, we report CIE where despite the use of iodixanol, the patient developed serious complications such as status epilepticus and hemiplegia after secondary coiling. The potential predisposing risk factors are also discussed.
CASE REPORT
A 66-year-old woman was admitted for the treatment of recanalization of a left middle cerebral artery (MCA) aneurysm. Ten years earlier, she underwent surgical clipping of an incidentally discovered MCA unruptured aneurysm. However, one year earlier, she presented with subarachnoid hemorrhage (SAH) following the rupture of the previously clipped MCA aneurysm (Fig. 1A) and underwent stent-assisted coiling at another hospital. The procedure was performed using 150 mL of iopromide (Ultravist®; Bayer Schering Pharma AG, Berlin, Germany) without adverse reaction to the contrast medium used.

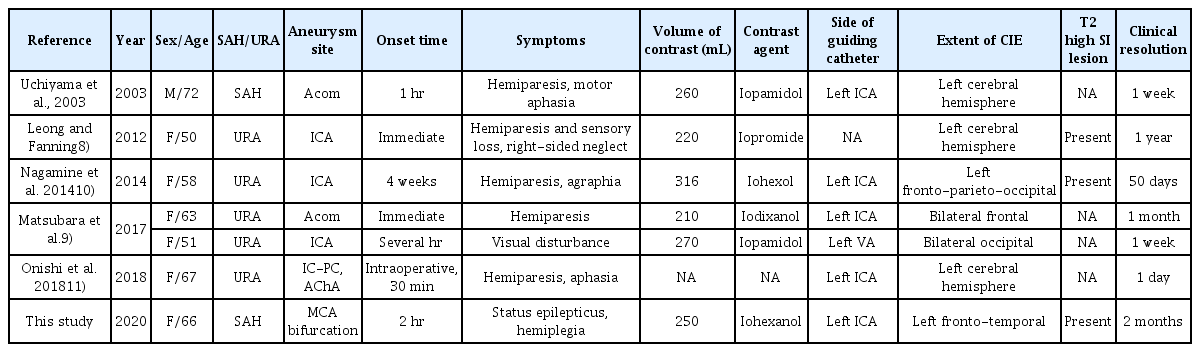
Clinical history before secondary coil embolization. (A) One year earlier, the patient had subarachnoid hemorrhage due to the rupture of the left middle cerebral artery aneurysm, which had previously undergone surgical clipping 10 years earlier. (B, C) Recent follow-up computed tomography before secondary coiling revealed encephalomalacia in the left frontotemporal area. (D, E) Recent cerebral angiography revealed major recanalization of the previously occluded aneurysm by surgical clipping and stent-assisted coiling.
Follow-up brain computed tomography (CT) revealed encephalomalacic change in the left frontal and temporal lobe caused by aneurysmal rupture (Fig. 1B, 1C). Cerebral angiography was performed using 100 mL of iodixanol (Visipaque 270; GE Healthcare, Princeton, NJ, USA) without any complications and revealed the major recanalization of previously coiled aneurysm (Fig. 1D, 1E). The coil embolization procedure was performed via the right transfemoral approach. A guidewire was inserted into the left internal carotid artery (ICA) and extended to the tortuous carotid artery. Subsequently, 6-Fr guiding catheters (Asahi Fubuki®; Asahi Intecc Co., Ltd., Aichi, Japan) were placed in the left ICA, and a microcatheter (SL 10; Stryker Neurovascular, Fremont, CA, USA) and microwire (Traxcess; Microvention, Tustin, CA, USA) were placed in the aneurysm sac through the strut of the previous stent (trans-cell technique). The aneurysm was completely obliterated with six coils (Axium Prime; Medtronic, Irvine, CA, USA), maintaining the distal MCA arterial flow. A total of 250 mL of iodixanol was used during the procedure. There was no thromboembolic event during the procedure. She recovered from anesthesia without any complications. However, the patient’s postoperative brain CT revealed contrast enhancement and gyral effacement, indicating cerebral edema in the left hemisphere (Fig. 2A).
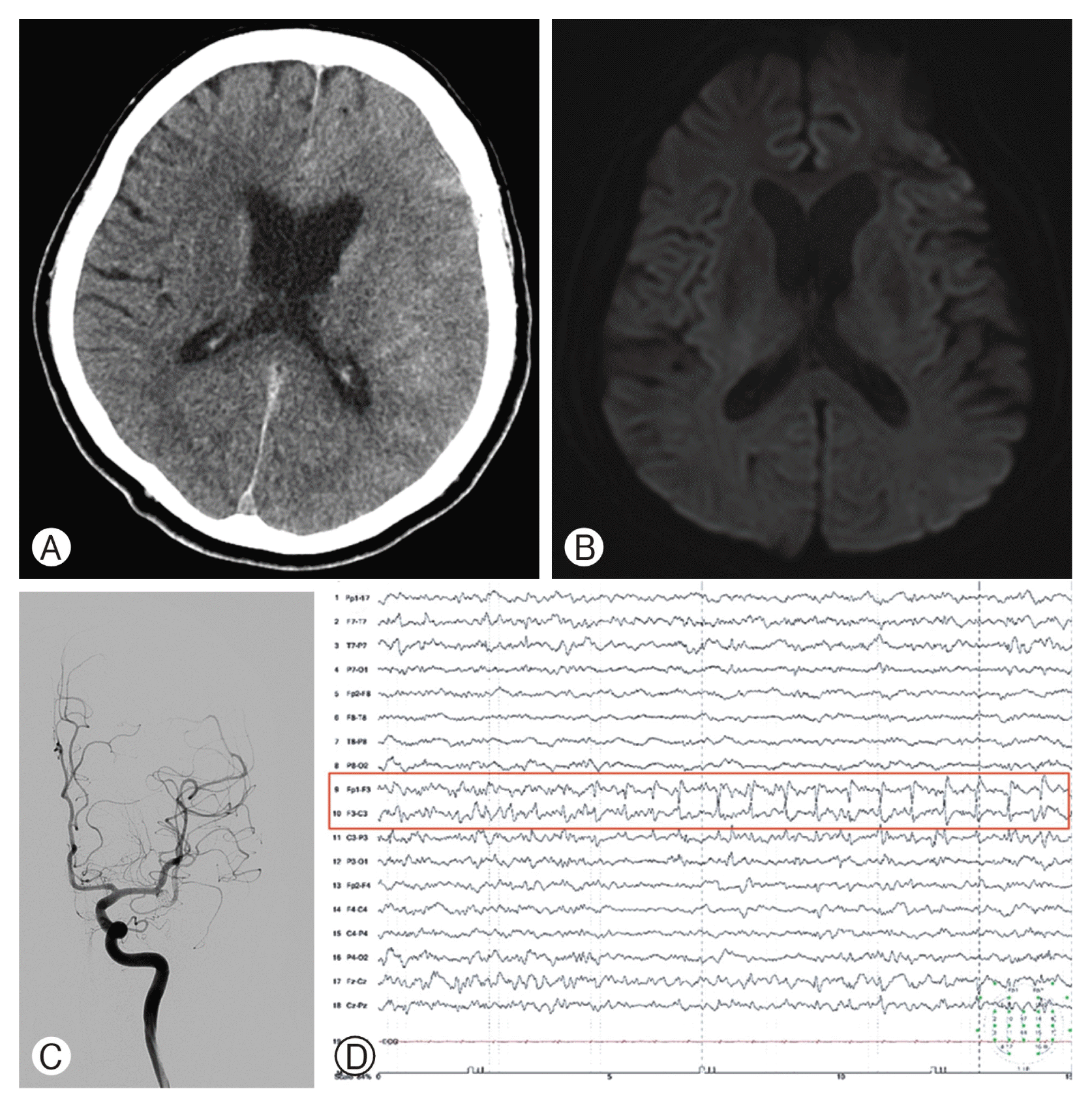
Postoperative findings. (A) Immediate postoperative computed tomography demonstrated contrast enhancement and gyral effacement extensively in the left hemisphere. (B) Postictal diffusion-weighted imaging revealed no evidence of acute infarction. (C) Follow-up of the left carotid angiogram 3 hr after secondary coiling confirmed the patent flow of the left middle artery and complete obliteration of the aneurysm. (D) Ictal electroencephalography revealed repetitive sharp waves concentrated in the left fronto-centro-parietal area (red box).
Two hr after the procedure, the patient developed sudden-onset right-sided hemiplegia and aphasia. She gradually became more lethargic and finally developed generalized tonic-clonic seizures for 5 min. Although intravenous lorazepam (4 mg) was administered repeatedly, the seizures did not stop and even prolonged for more than 30 min. She was administered 1,000-mg phenytoin sodium followed by 1,000-mg sodium valproate with no respite in seizures. She was considered to be experiencing refractory status epilepticus and moved to the intensive care unit for therapeutic coma induction. Seizure control was achieved at a dose of 2 mg/kg/hr of propofol. Under high suspicion of postoperative stroke, a follow-up cerebral angiogram and diffusion-weighted imaging were performed, but they revealed patent MCA flow without cerebral infarction (Fig. 2B, 2C). Electroencephalography demonstrated repetitive sharp waves concentrated in the left frontocentroparietal area (Fig. 2D). Hydration and intravenous administration of methylprednisolone, mannitol, and anticonvulsant agent were also provided for symptom control. Propofol tapering was attempted after 72 hr of seizure-free period.
Recovery (with spontaneous eye-opening) started to occur on postoperative day7), and the hemiplegia gradually resolved. T2-weighted images demonstrated extensive cerebral edema of the left fronto-temporoparietal area (Fig. 3A, 3B). Two-month follow-up magnetic resonance imaging (MRI) revealed that the edema markedly improved in the parietal area but remained in the frontal area (Fig. 3C, 3D). Finally, the patient completely recovered without any neurologic sequelae.
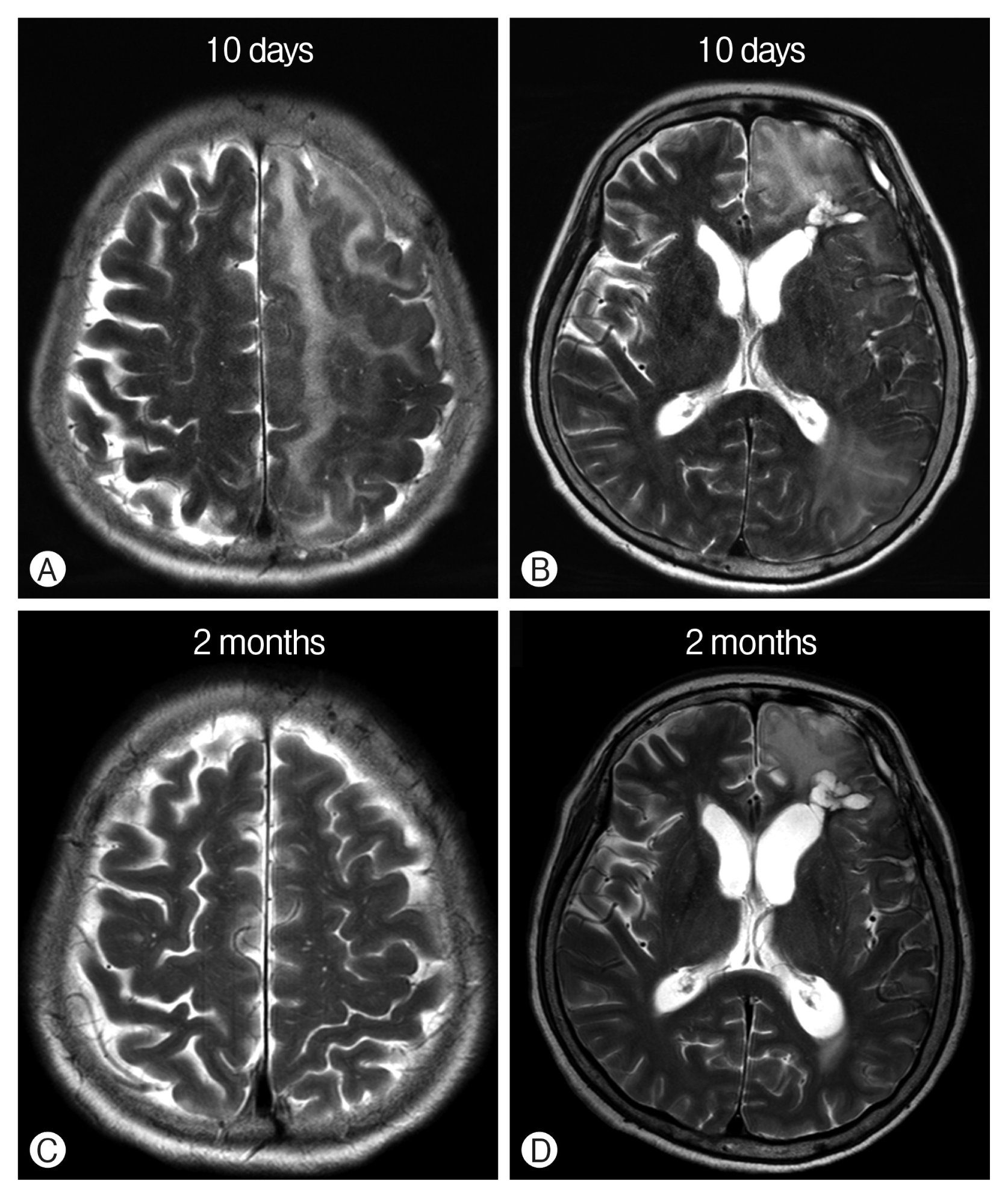
Serial postoperative magnetic resonance images. (A, B) One-week postoperative T2-weighted images (WI) demonstrated extensive involvement of contrast-induced encephalopathy in the left hemisphere. (C, D) Two-month postoperative T2-WI showed that high signal lesion in the parietal area was no longer observed, but that in the frontal area adjacent to the encephalomalacic lesion remained.
DISCUSSION
Since 2010, there has been an increasing trend in the incidence of CIE (Table 1). To the best of our knowledge, there has been only one case that reports CIE after coil embolization using iodixanol contrast medium9).
The clinical presentation of CIE can sometimes mimic acute stroke, but imaging findings present several characteristics of CIE. The most common symptoms of CIE include cortical blindness, headache, vomiting, agitation, hemiplegia, seizures, confusion, transient global amnesia, and dysarthria–aphasia4,17). Cerebral angiography and diffuse-weighted imaging were also performed because of high clinical suspicion for postoperative acute ischemic stroke. Radiologic findings of CIE are characterized, consistent with our case, by gyral swelling and hyperintensity on T2 image on MRI and also abnormal cortical contrast enhancement, edema, and subarachnoid contrast enhancement on CT8). Thus, it is important to differentiate between CIE and stroke as promptly and accurately as possible.
Possible risk factors for CIE include chronic hypertension (HTN) with impaired cerebral autoregulation, chronic kidney disease, and a large volume of contrast media9,17). This patient had a history of chronic HTN, previous SAH, and the large amount of contrast (250 mL) administration for repeat arteriograms. Additionally, given the evidence of long-standing persistent T2 high signal of the left frontal lobe in the late stage of CIE, the presence of precedent encephalomalacia may have predisposed the patients to CIE and facilitated the contrast agent to disrupt the blood-brain barrier, causing cerebral edema5).
It is unclear whether the difference in types and doses of the contrast media develops CIE. Theoretically, iodixanol, an iso-osmolar contrast agent, hardly induces blood-brain barrier damage, and it has been associated with fewer adverse effects and only rarely with CIE7,12). A comparison study showed both iodixanol and iopromide to be safe, effective, and well-tolerated14). CIE also developed during angiography procedure with contrast volumes less than 40 mL and even with 25 mL of contrast media for local injection into the carotid artery6,13). In our case, CIE did not develop during cerebral angiography with 100 mL of iodixanol but during coil embolization with 250 mL of iodixanol. Thus, iodixanol-induced encephalopathy appears to be an idiosyncratic reaction.
There is no definite treatment strategy of CIE given the unexpected and rare nature of the complication.8) Previous reports demonstrated that hydration and intravenous steroid and mannitol administration might help symptom control7,12). Although the prognosis of CIE has been benign with a wait-and-see approach in most reported cases12), CIE could present with severe neurological symptoms, and permanent neurological symptoms may remain8).
CONCLUSION
Despite the use of iodixanol contrast media, CIE can develop during coil embolization. Endovascular neurosurgeons should always be aware of CIE, especially when encephalomalacia is present.
Notes
No potential conflict of interest relevant to this article was reported.