Lumbar Calcifying Pseudoneoplasm as a Rare Cause of Cauda Equina Syndrome: A Case Report
Article information
Abstract
A calcifying pseudo-tumor of the spine, also called calcifying pseudoneoplasm of the neuraxis (CAPNON), is a rare non-neoplastic lesion that can occur anywhere in the central nervous system. Although CAPNON shows tumor-like behavior, it is believed to be benign and of inflammatory-reactive origin. It may cause many neurologic symptoms by compressing adjacent structures. We report the case of a 45-year-old man who presented with cauda equina syndrome. Magnetic resonance imaging revealed a partially calcified mass compressing the spinal cord. Gross total resection was carried out and the patient’s symptoms improved. Histopathology proved that the mass was CAPNON. Although spinal localization of CAPNON is rare, we should know and recognize this entity based on appropriate imaging findings because radical excision has an excellent prognosis.
INTRODUCTION
Calcifying fibrous pseudoneoplasm, also known as calcifying pseudoneoplasm of the neuraxis (CAPNON) when found in the central nervous system, are rare pathological entities that are benign non- neoplastic fibrous lesions1,9,10,19). It’s usually found in patients who are above 50 years and, in some cases, is associated with neurofibromatosis type 24,5,14). The disease may be found in the peritoneum, neck, mediastinum, breast, extremities, central nervous system, and spinal cord11,12). When the spine is involved, its most frequently located in cervical region followed by thoracic and lumber areas. CAPNON may be both intra- and extra-dural16). Pathogenesis of CAPNON is still unclear, however, it is thought of as reactive lesions with abundant hyalinized collagenous fibrous tissue with psammomatous or dystrophic calcification and focal lymphoplasmacytic infiltrate3,7,13).
Patients who are diagnosed with CAPNON can have a variety of symptoms from no symptoms at all to myelopathies, depending on the location and compression of the surrounding neural structure9,15). Therefore, the management of CAPNON is largely dependent on the symptoms, and when patients show severe neurologic deficits, complete surgical removal is the treatment of choice18). Since CAPNON has excellent treatment outcomes when successfully removed, a thorough diagnostic screening needs to be carried out. Magnetic resonance imaging (MRI) is the modality of choice of investigation which shows hypo intense T1- and T2-weighted images with limited edema and contrast enhancement13,16,17).
We present a rare case of CAPNON in the lumbosacral region showing cauda equine syndrome, mimicking hourglass neurinoma or ependymoma.
CASE REPORT
A 45-year-old man came to the hospital emergency room (ER) with severe low back pain and motor weakness in both low extremities for 5 days (motor grade 3). Neurologic examination revealed altered sensations in his buttocks, inner thighs, backs of legs and feet, typically demonstrating saddle-like paresthesia in L5-S1 dermatome. He had also altered bowel and bladder habits prior to the visit to the ER. His overall symptoms suggested the possibility of cauda equina syndrome which could lead to a possible emergency operation and thus, was admitted to our neurosurgery department for further diagnostic evaluations.
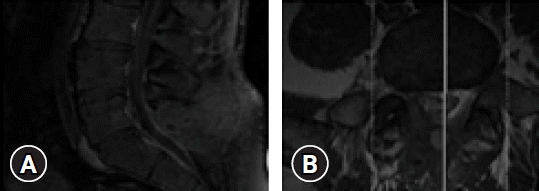
MRI showed an intradural mass extending into the neural foramen where signals revealed partial calcification at the L5-S1 disc level (Fig. 1A, B). Surgical removal was carried out via laminectomy of L5 lamina to achieve clear access to intradural calcified lesions. The dura was incised vertically just above the mass. The incision was then extended just above and below the lesion to get a clear view of the tumor. The tumor was resting on the posterior dura, adhered with the fibers of S1 within the neural foramen.

(A) Preoperative sagittal T1-weighted magnetic resonance imaging (MRI) with gadolinium enhancement displaying the L5-S1 intradural neoplasm. (B) Preoperative axial T1-weighted MRI with gadolinium enhancement displaying the L5-S1 intradural neoplasm.
During surgery, the nerve roots of the dural sac adjacent to the tumor were moved by gentle spatulation with a small dissector. The lesion had a fibrous consistency due to the presence of calcifications. There were no hemorrhagic focus and infiltration into the surrounding tissues. The mass was successfully removed without giving any injury to surrounding neurologic anatomy.
A biopsy was carried out with the mass acquired during surgery. It confirmed the diagnosis of CAPNON. The histopathological slide was stained for hematoxylin and eosin which showed positivity for the expression of factor XIIIa and vimentin and negativity for the expression of CD31, CD34, anaplastic lymphoma kinase (ALK)-1, smooth muscle actin (SMA), protein S100 and VIII factor (Fig. 2A, B).
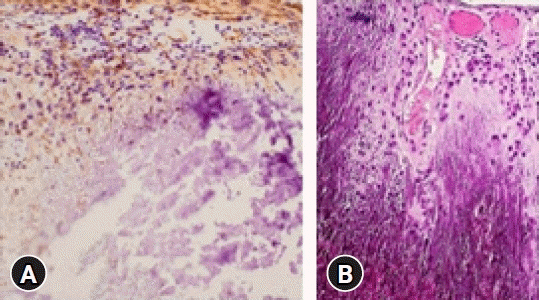
(A) Histological findings include central calcification with surrounding chondroid matrix organized in a nodular pattern. There are palisade spindle cells around the nodules (hematoxylin and eosin stain [H&E], ×100 magnification). (B) Histological findings showing inflammatory cells in the fibrovascular stroma between chondromyxoid cells.
On the first day after surgery the patient showed complete resolution of cauda syndrome. After three months, we performed a follow-up lumbosacral MRI with contrast which showed no recurrence (Fig. 3A, B). After 15 months, patients came for follow-up with no signs and symptoms of any recurrent disease.
DISCUSSION
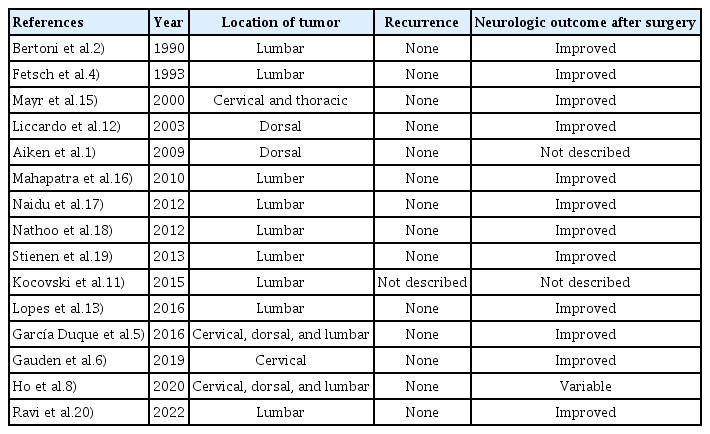
Calcifying fibrous pseudoneoplams occurring in the neuraxis is very rare. Nevertheless, it is rather more common for it to occur in the spine than in the brain6,7). To the author’s knowledge, there are only 29 similar cases of CAPNON occurring in the spine so far (Table 1). According to previous studies, the tumor has a male preference occurring mostly in patients aging more than fifty and can be present in all levels of the spine3). The characteristics of the tumor also varied greatly from case to case, where it was interosseous, intradural, extradural and even intradural extramedullary8,16).
CAPNON can only be diagnosed after a full histopathologic and immunohistochemical analysis4,14,19). It is difficult to suspect and diagnose the disease with only conventional imaging workup such as MRI, computed tomography (CT) scans and X-rays. The tumor usually displays partial calcifications on CT scans and on the MRI it usually shows a well-defined hypointense lesion in T1 and T2 weighted images along with limited hyperintensity after contrast enhancement T1 weighted imaging13,15,20). Due to these non-specific characteristics of the tumor, it is crucial for physicians to rule out other more common possible disease entities. Chondromas, chordomas, meningiomas and schwannomas were considered in the differential diagnosis for extra-axial localizations, while oligodendrogliomas and ganglioglioma for intra-axial localizations3,12).
The typical histopathological characteristics of CAPNON are predominantly lymphoplasmacytic infiltrate with abundant hyalinized collagen, dystrophic calcifications, fibrous stroma with chondromixoid matrix, epithelioid cells arranged in palisades and similar inflammatory reaction with giant cells3,7,16).
There have been some controversies regarding CAPNON being inflammatory or neoplastic. Pathologists had hypothesized that calcifying fibrous pseudotumor, as a slow-growing benign tumor, derived from inflammatory myofibroblastic tumor than from a typically inflammatory lesion3). The anatomopathologists in recent years have hypothesized that calcifying fibrous pseudotumor was true benign tumor entities without having an inflammatory origin because they did not present the characteristic immunohistochemical markers of inflammatory myofibroblastic tumors2,14).
In the case presented in this study it showed similar results where it showed hyalinized leiomyoma (positive for smooth muscle markers, SMA, desmin, negative for the expression of factor XIII); Inflammatory myofibroblastic tumor (negative for SMA expression and only sporadically positive for Keratin and desmin expression, while always positive for the expression of factor XIIIa and ALK).
The overall treatment outcome is known to be excellent with complete surgical removal7). Recurrences appeared rare even after complete or incomplete resection of the disease3,7). Stienen et al.19) found no recurrence of lesion in 92.3% of the cases.
CONCLUSION
CAPNON is a rare but benign tumor now considered an autonomous anatomopathological entity. Neurosurgeons should be aware of the disease and recognize this entity through conventional image workups and histopathologic outcomes because radical excision determines an excellent prognosis.
Notes
No potential conflict of interest relevant to this article was reported.