INTRODUCTION
Ependymoma is a glial tumor that arises from the ependyma, a tissue of the central nervous system. Ependymomas generally present as centrally situated intramedullary tumors within the thoracic/cervico-thoracic cord7). Ependymal cells spring from primitive progenitor, the ependymoglia cell, also called “tanycyte”. On tracing ependymal and progenital development, transitional types of ependymomas exist. On the World Health Organization (WHO) classification, ependymomas are divided in three major groups: subependymoma (WHO grade I), myxopapillary ependymoma (WHO grade I), ependymoma (WHO grade II), and anaplastic ependymoma (WHO grade III). Conventional (WHO grade II) ependymomas, includes the tanycytic variant.
Tanycytic ependymoma (TE) is a WHO grade II tumor with histological characteristics distinct from the typical features of commonly encountered ependymomas4). TE is one of the transitional types of ependymoma2). The most common symptoms of TE is the gradual deterioration of motor functions and/or sensory disturbances, such as heaviness, numbness, and weakness of the low back and lower extremities including the waist (paraparesis) accompanied with gait disturbance and urinary incontinence. TE has specific molecular structure. The classic ependymal rosettes and perivascular pseudorosettes of ependymoma are replaced by more fibrillar, spindle cells of tanycyte. The cellular appearance and arrangement may mimic other tumors such as schwannoma and astrocytoma, which makes the diagnosis challenging and hence warrants further evaluation. Since the first case of Friede and Pollak2) was reported in 1978, only 34 cases have been reported by 2016. Herein, we present four cases (Table 1) of rare ependymoma, TE, which were initially thought to be schwannoma or astrocytoma.
CASE REPORTS
1. Case 1
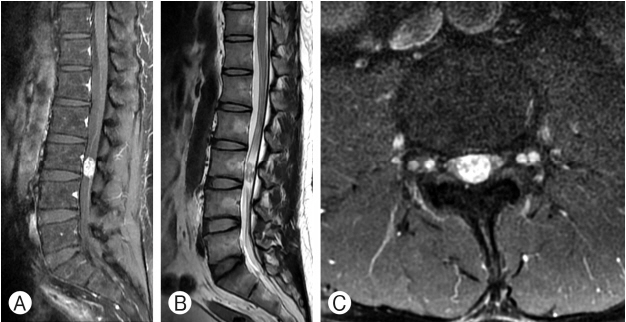
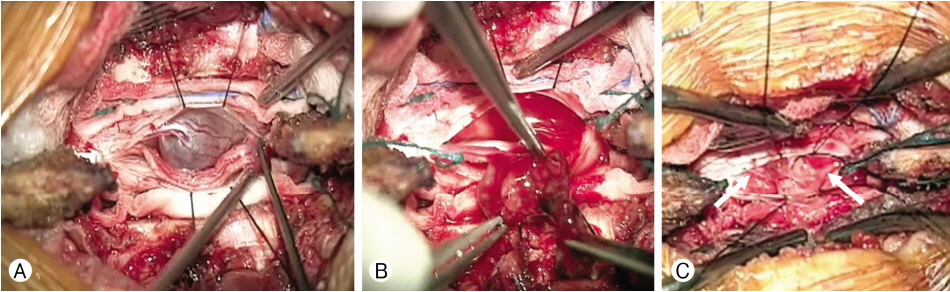
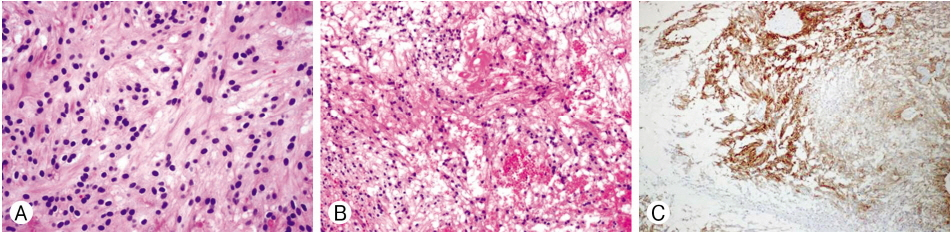
A 45-year-old man in good health had been having issues with pain in both lower legs and constipation since December 2014. He went to a local clinic and had a magnetic resonance imaging (MRI) done to rule out spinal stenosis. He was referred to our neurosurgery clinic after MRI of the lumbar spine which revealed an extramedullary lesion at the L3 level. His neurologic examination showed that he had visual analog scale 8 of pain on both legs involving the posterior thigh and calves along the L4-L5 dermatomal distributions. The patient also noted bowel dysfunction. There was no evidence of abnormal reflexes and motor weakness. On MRI, the tumor measured 1.0×1.1×1.8 cm, which was at the L3 level. It was an oval-shaped intradural extramedullary filum terminale mass lesion, avidly enhancing on T1-weighted imaging (WI) with mixed intensity on T2-WI (Fig. 1A-C). A laminectomy at L3 was performed. Fusiform-shaped spinal cord tumor that originated from L5 or S1 on the right side was exposed. To preserve the nerve root, sub-capsular dissection and tumor removal were done. The tumor capsule with nerve root was preserved (Fig. 2A-C). Microscopically, the tumor was composed of spindle cells with a palisading pattern that resembles verocay bodies of schwannoma and clear cells in some areas (Fig. 3A). Moreover, prominent hyaline blood vessels, macrophages, and few giant cells were detected (Fig. 3B). The tumor cells where strongly and uniformly positive for glial fribillary acidic protein (GFAP) (Fig. 3C). These findings support the diagnosis of TE. Because gross tumor removal (GTR) of an ependymoma was achieved, no adjuvant therapy was implemented. The patient’s postoperative course was uneventful. He experienced improvement in his both lower extremity pain. Postoperative MRI showed complete resection with no evidence for recurrence (Fig. 4A, B). Four years after surgery, the patient is free from recurrence.
2. Case 2
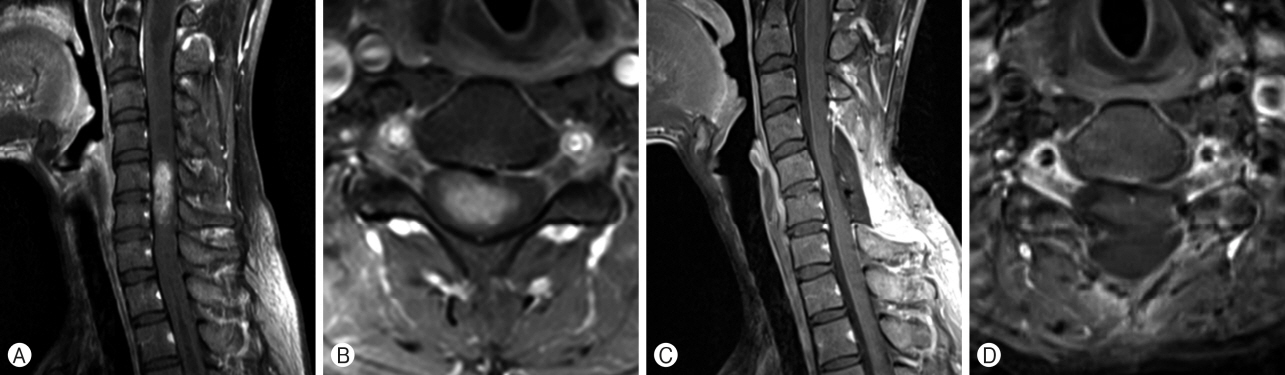
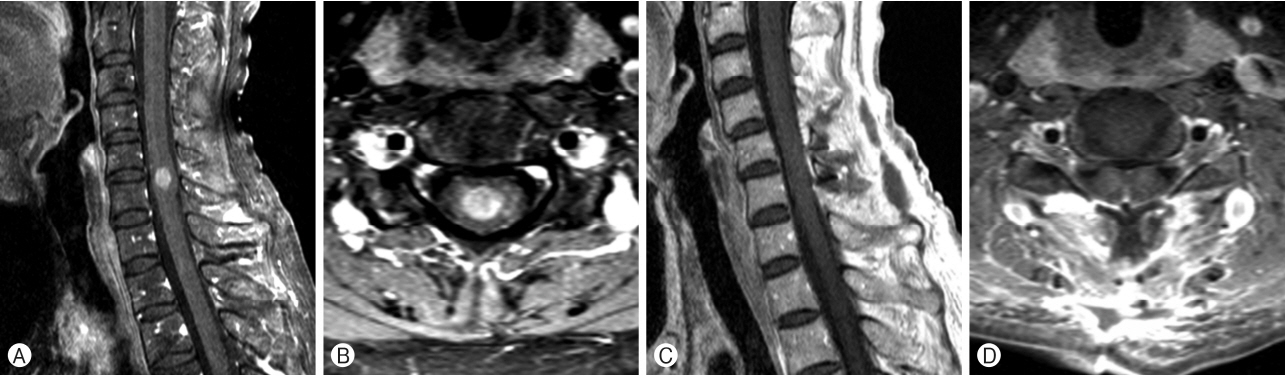
A 44-year-old woman with no medical problem came to visit us for numbness and tingling sensation in right forearm since 2009. On MRI, the tumor measured 3.0×1.2×1.6 cm, which was at the C5 level. It was enhancing on T1-WI (Fig. 5A, B). C5 laminectomy was performed, and GTR was done. Microscopically, the tumor was diagnosed with TE. After the surgery, no adjuvant therapy was planned. Postoperative MRI showed complete resection (Fig. 5C, D). Eight years after surgery, the patient has no evidence of recurrence.
3. Case 3
A 59-year-old woman with old tuberculosis and major depressive disorder was referred for right arm pain and myelopathy. She had no motor weakness but hypoesthesia at the right arm. On MRI, the tumor measured 3.0×1.2×1.6 cm, which was at the C5 level (Fig. 6A, B). C5 laminectomy was performed, and GTR was done. The pathologist confirmed that the tumor was TE. After tumor removal, no adjuvant therapy was performed. Postoperative MRI showed complete resection (Fig. 6C, D). Seven years after surgery, the patient is free from recurrence.
4. Case 4
A 41-year-old woman in good health had been having issues with pain on both legs. She had no motor weakness. On MRI, the tumor measured 1.2×0.7×0.7 cm, which was at the L2 level. It was extramedullary mass lesion, enhancing on T1-WI (Fig. 7A, B). L2 laminectomy was performed. The tumor was attached to the filum terminale. The root was sacrificed, and the tumor was completely removed. After the surgery, no neurologic deficit was found. The tumor was confirmed to be TE. Postoperative MRI showed complete resection (Fig. 7C, D). Four years after surgery, the patient is free from recurrence.
DISCUSSION
TE is of low-to-moderate cellularity characterized by a flow of elongated cells with nuclear pleomorphism. Mitotic figures are usually absent. It has inconspicuous features that are not typical of ependymoma. The distinctive perivascular pseudorosette pattern of ependymoma cells is infrequently encountered3). Their nuclei are oval to spindle shaped with evident pleomorphism. The cell processes are not fluffy as with the cellular type of ependymoma8). Radiologically, ependymoma is almost always intramedullary. In our cases the tumor was an extramedullary lesion that also avidly enhanced on T1-WI with gadolinium contrast enhancement. Pilocytic astrocytomas are rarely enhanced11). TE cells may mimic other tumor cells with similar features under light microscopy often lending to a challenging diagnosis3,5,6,9). The spindle appearance of the cells, for instance, is similar to that of schwannoma cells. TE stains positive for GFAP and vimentin but rarely stains for S-100. This helps to differentiate it from schwannoma, which is S-100 positive and GFAP negative. Although pilocytic astrocytoma is GFAP positive, it almost never stains for vimentin, which helps to discriminate it from TE (Table 2)5), Pilocytic astrocytoma also presents Rosenthal fibers and eosinophilic granular bodies1,7). Recently, Tao et al.13) reported 40 cases of TE in 2017. Approximately 4000 spinal cord tumors were resected surgically, of which 40 were identified with TE. In terms of prognosis, at a median follow-up of 43 (range, 1-101) months, 1- and 5-year progression-free survival rates were 100.0% and 97.5%, respectively13). The current management is radical excision of spinal TE. It is unlikely to recur if the tumor is completely removed12). Repeat resection or radiation treatment will be recommended in the event of recurrence10). Adjuvant chemotherapy is not recommended14). Through this paper, we reported four cases of TE diagnosed in Korea. Until now, only one case was reported in Korea6). Our histologically confirmed four cases will contribute greatly to research on TE in Korea. We also hope these Korean cases will be used as valuable material for interracial and intercontinental study of TE.
CONCLUSION
Rare cases of spinal TE were presented. TEs are an uncommon fibrillary variant of ependymomas with typical pathological characteristics. A careful and detailed histological inspection with utilization of immunohistochemical stains and ultrastructural microscopy may be necessary to distinguish TE from other neoplasms such as schwannoma and pilocytic ependymoma. We believe that this paper could help in the accurate diagnosis and giving appropriate treatment to such population.