INTRODUCTION
Cervical disc arthroplasty (CDA) is a viable and effective treatment of cervical degenerative disc diseases and has been proposed as an alternative to the conventional anterior cervical discectomy and fusion. The formation of heterotopic ossification (HO) is considered one of the major complications after CDA, which, in the setting of CDA, specifically refers to the undesirable ectopic osteogenesis anterior, posterior, or lateral to the implant and subsequently plays a contradictory role against the fundamental goal of CDA by impeding the range of motion (ROM) of the implanted devices2).
While the majority of studies have focused on the identifications of etiology, risk factors of HO as well as their feasible prevention measures, the question that might be a crucial concern to all surgeons is whether the presence of HO would pose a significant impact on the clinical outcomes has not been properly addressed currently. Moreover, scarce reference could be searched among the past published literatures reporting the accumulation of this HO rear to the previously inserted, moving implant and consequently incurring an unexpected cervical spondylotic myelopathy (CSM)16). This case report describes a patient presented with clinical signs of cervical myelopathy caused by the atypical HO rear to the still functioning cervical artificial disc implant.
CASE REPORT
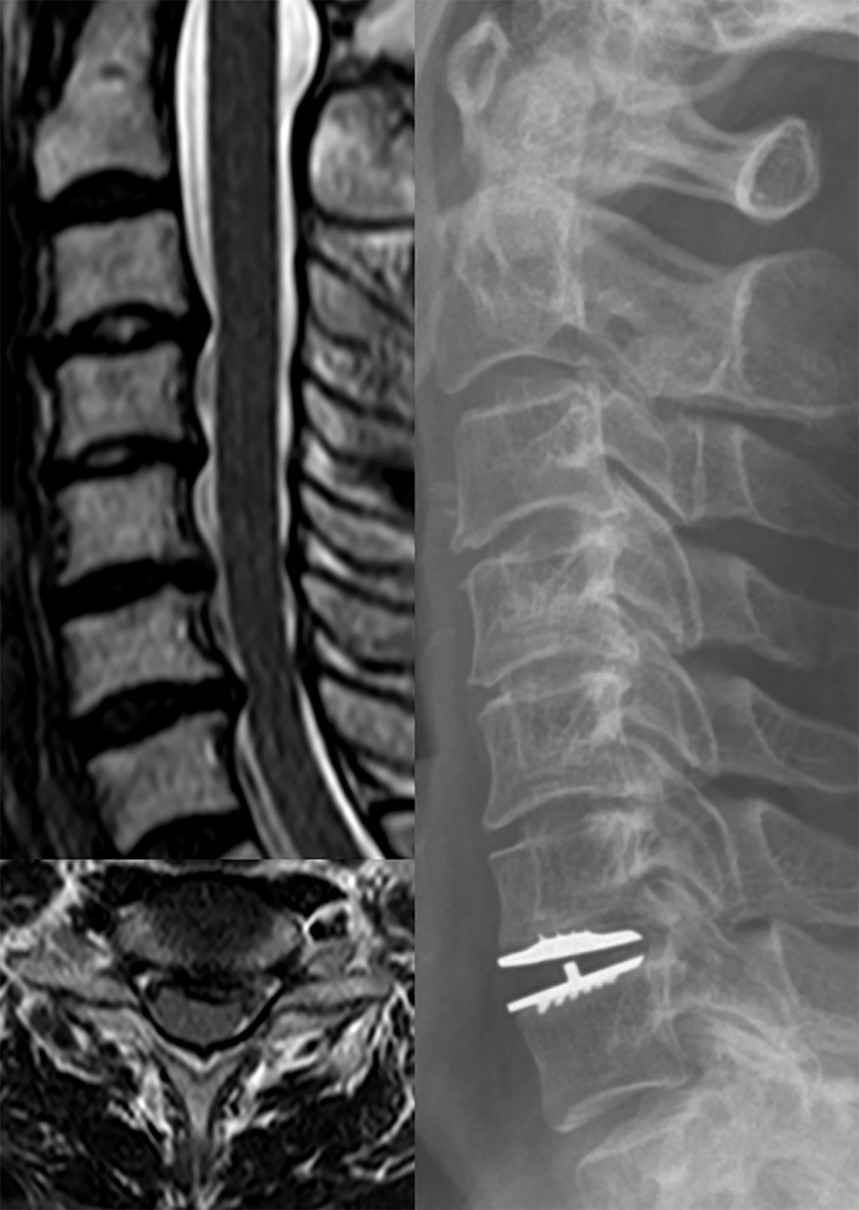
This 63-year-old female presented with the clinical signs of myelopathy almost 5 years after the initial implantation of a Mobi-C disc prosthesis (Zimmer Biomet, Warsaw, IN, USA) at the C6-7 level due to the left side upper extremity radiculopathy affected with both spinal canal and foraminal stenosis from a local hospital (Fig. 1). Started from approximately a month prior to the referral, an intractable, lancinating pain combined with electricity like paresthesia, especially at the region of the left elbow as well as both palm and fingertips hindered her from the indoor activity at the kitchen, which later rapidly prevailed to refrain her from the outdoor activity due to gait disturbance by paraparesis progression. She showed the dominant radiculopathy and paresthesia at the left C6 and C7 dermatome exaggerated response of both the biceps and triceps brachial reflex, and grade 4 weakness of both lower extremities. The new magnetic resonance imaging on admission surprisingly demonstrated another severe spinal cord compression and consequent swelling and signal change inside the cord at the previously addressed level of C6-7, which was attributed to the bony spurs collections rear to the Mobi-C disc prosthesis as manifested on the concomitant computed tomography scan (Fig. 2).
1. Initial Surgery and Its Consequence
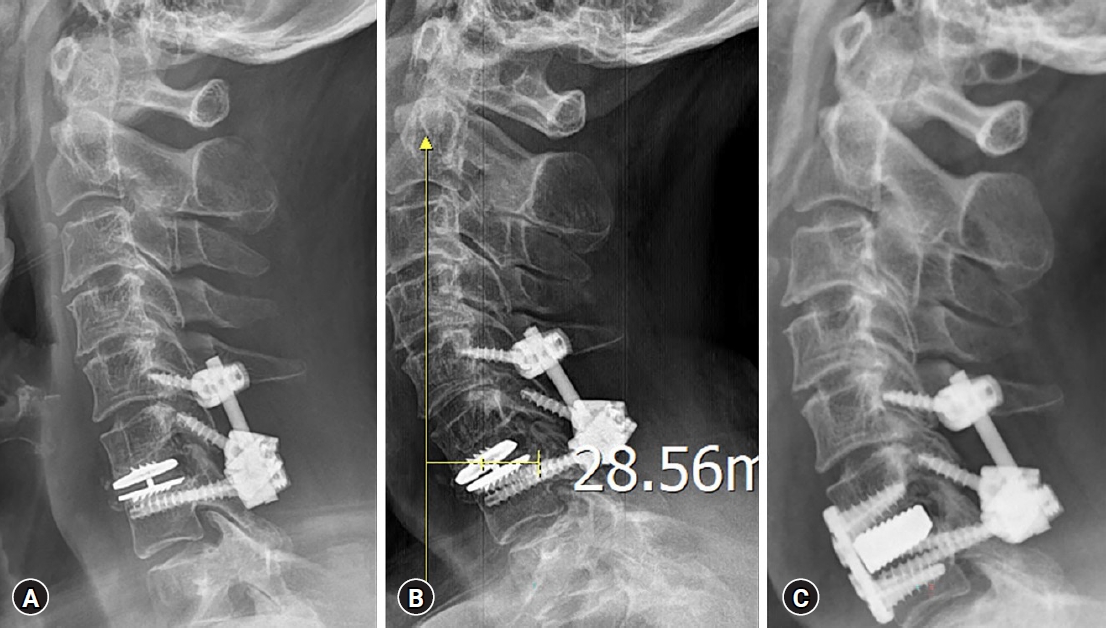
In order to fully decompress the affected spinal cord as well as to offer initial stabilization to the still functioning prosthesis, a posterior full decompressive laminectomy added with C6 lateral mass and C7 pedicle screw fixation was performed. This was originally schemed to avoid any untoward complication occurring during the revision approach or manipulation to resect off the HO that might stick to the ventral aspect of the dura. Upon gentle retraction of the lateral margin of the dura at the axillar portion of the exiting C7 root, the surgeon could directly encounter the expected aberrant, discolored but protruded bony spur mass ventrally affecting the cervical spinal cord (Fig. 3). Dorsal cortical bone over the left side C6 lateral mass had breakage violation during the screw insertion due to simple technical error, which eventually obliged the surgeon to shift the fixation level to the cranial level of C5, unfortunately (Fig. 4A).
Fortunately, the patient showed prompt recovery from her original neurological defect upon initial procedure, with a trace of neuropathic component remnant at the distal part of the upper limb such as fingertip.
2. Second Surgical Procedure and Postoperative Course
The patient managed to be in fair condition, clinically and radiologically, up to two months after the initial procedure till she started to appeal the involuntarily, ‘forwardly tilted neck’ condition on the second visit to the outpatient office. The additional radiograph on lateral posture featured the well-maintained posterior fixation constructs while a concomitantly veered, exaggeratedly flexed but not dislocated moving core inside the Mobi-C artificial disc even during the neutral standing posture with the sagittal vertical axis distance exceeding more than 28 mm (Fig. 4B). Due to the constant discomfort caused by elongated, curved neck posture with subsequent incapability to maintain the proper horizontal gaze, she asked for another surgical corrective measure to overcome this unexpected, deformed status of twice treated neck. The flexed but still functioning prosthesis, as well as the ectopic bone deposited rear to the device for the sake of additional direct decompression of the spinal cord, had to be removed and switched to a firm anterior fusion arthrodesis to provide an immediate correction to the cervical kyphotic curvature change (Fig. 4C).
She also managed to recover fairly even after this second revision surgery and was discharged a few days later leaving a specific expression that describes her condition; she can now ‘look up’ her family without the necessity of rigid external orthosis.
DISCUSSION
HO is defined as the formation of bone outside the skeletal system. It is a well-known phenomenon that might disable patients in total hip or knee joint replacement5,8), but it has also been repeatedly detected in spinal patients after the wide acceptance of CDA. Intraoperative measures to deter the delayed HO development after cervical artificial disc implantation involve a gentle surgical technique, avoidance of muscle trauma, and repetitive rinsing of the surgical field14). Selection of the largest possible caliber for the contacting plate part of the CDA device that might be capable to cover the majority of the surface area for the opened endplates (adequacy of the endplate coverage) could also play a deterrent role against this HO progression by minimizing the unnoticeable discharges from the both above and below vertebral body components11-13). Despite all these efforts, the meta-analysis pooled data from the Kong present the 53.6% and 47.5% prevalence of HO and severe HO respectively within 5 to 10 years after CDA with its positive correlation with the length of follow-up period6).
Although the HO might occur in a substantial proportion of CDA switched spinal segments, but does not appear to lead to a decline in clinical outcomes. Its incidence, progression, clinical Implications, and risk factors based on the long-term outcomes up to seven years have been reported after the usage of Mobi-C device and the trial results have been consistent with the continued non-inferiority of CDA for clinical outcomes and lower cumulative reoperation rates despite up to 26.6% of its incidence3,7). Solid bridging of spurs usually remains asymptomatic and is predominantly discovered at the anterior vertebral edges of the treated interspace merely through routine follow-up radiological examinations1,7,13,15). Some studies demonstrated that the changes in biomechanical factors were associated with the prevalence and/or severity of HO; however, a causal relationship between these factors remains unproven. Hu et al.4) have iterated that a more than 5° increase in immediate post-operative disc space angle and less segmental ROM have conferred a negative effect on HO formation. Meanwhile, Shen et al.10) have previously concluded that endplate coverage of less than 93.8% or intervertebral height change of more than 1.8 mm would exacerbate the non-uniform distribution of stress in the bone-implant interface and promote posterior HO development after cervical disc replacement. Further investigations are warranted to corroborate these risk factors, including multilevel calcified disc herniation, severe spondylosis, and suboptimal placement of the device during primary cervical disc replacement surgery.
Consequently, this unprecedented HO collection rear to the device as well as into the spinal canal eventually incurring the clinical CSM neurologic feature might not have been discovered or anticipated neither by the surgeon or patient herself, although she has been followed up with regular functional radiographs after initial procedure. The authors might suggest the conventional hypothesis that permanent micro stress between the bony endplate and the device, which might have originated from the non-physiologic prosthesis motion, might have promoted osseous spur formation and its enlargement2). Probably an untoward synergy between the non-physiological motions created from the ventrally placed prosthesis with relatively smaller caliber along with the presumably insufficiently resected, remnant dorsal osteophytes might have triggered this appositional bone growth over the residual osteophytes, whose growths are well informed to be promoted by the untoward segmental motion. But there could be no clear elucidation regarding its isolated collection rear to the initial implant, sparing the preferential ventral aspect of the addressed cervical level. Moreover, this presumably consolidated, functionally deprived prosthesis after the secondary posterior fixation surgery was not fully immobilized, retaining its inherent mobility to be flexed. This post-surgical phenomenon suggests the lack of capability for both the posteriorly sealed HO as well as posterior stand-alone fixation to provide firm stability over the functionally deprived cervical motion segment after the untoward artificial disc insertion. As iterated above, a conversion to the solid anterior fusion after full removal of the causative mobile device as well as the ectopic bone deposits would be the preferred revision measure over the simple posterior decompression and fixation for the sake of cervical curvature maintenance.
Recently, there has been data emerging to suggest that the formation of HO might be affected by an increased preoperative signal intensity in the spinal cord, postoperative ROM at the surgical level, and the prosthesis vertebral ratio9). However, it should be noted that, despite these hypotheses, there has been a paucity of consistent evidence. Larger scale investigations are needed to further corroborate the risk factors on the genesis of HO, especially for those lobulated rear to the implant causing CSM as in our case, after CDA.
CONCLUSION
To the author’s knowledge, such extensive bone accumulation posterior to a functional Mobi-C cervical prosthesis causing myelopathy has not yet been reported in the literature. Conversion to a solid anterior fusion after full removal of the causative device as well as the ectopic bone deposits would be preferred to simple posterior decompression for the sake of cervical curvature maintenance.