INTRODUCTION
The most typical reason for elderly patients aged 65 and above to undergo spinal surgery is lumbar spinal stenosis (LSS)15). If non-invasive treatment methods do not yield the desired results, the next step would be to seek surgical treatment for LSS. Surgical decompression is advised for individuals with neurogenic claudication, motor impairment, and a compromised spinal canal. The traditional method for decompressing the neural canal was wide laminectomy, which frequently required extensive removal of posterior supporting structures, including paraspinal muscles. However, this could result in instability or the syndrome of failed back surgery17). Biomechanical studies emphasize the crucial role of the posterior column, comprising paraspinal muscles, inter-and supra-spinous ligaments, and facet joints, in maintaining the stability of the spine1,16). Therefore, maintaining the posterior stabilizing structures is crucial for achieving good surgical results.
Minimally invasive (MI) techniques have been proposed for several decades to reduce damage to paraspinal muscles or posterior stabilizing structures. These techniques include the tubular retractor approach with microscopic or endoscopic assistance and various endoscopic approaches. Recently, a MI technique called unilateral biportal endoscopic (UBE) spinal surgery has been introduced4,8). The UBE technique involves dividing the operative field into two channels for viewing and working. The procedure is performed using conventional surgical instruments in a separate working channel under a clear, magnified endoscopic view in a water medium.
The purpose of this study is to provide a description of the surgical technique and the early clinical and radiological outcomes that resulted from the author's adoption of the UBE surgical technique for decompression of spinal stenosis.
MATERIALS AND METHODS
Between February 2019 and July 2021, 47 patients diagnosed with LSS underwent UBE. Prior to the procedure, standard clinical and radiological evaluations were conducted, and patients were followed up for up to 12 months. Before surgery, patients received at least 6 weeks of conservative treatment, which included pain medication, physical therapy, and nerve block. The majority of patients reported pain in their lower back, buttocks, and legs, as well as claudication. The diagnosis of LSS was confirmed using magnetic resonance imaging and/or computed tomography with 3-dimensional reconstruction. The surgical indication was neurogenic claudication or radiculopathy accompanied by LSS, refractory to conservative treatment for more than 6 weeks. Preoperative dynamic X-rays were also performed on all patients to detect significant spinal instability. This study included patients with one or two levels of LSS, as well as those with stenosis accompanied by a herniated disc or grade 1 degenerative spondylolisthesis. However, patients with high-grade spondylolisthesis and accompanying severe extraforaminal stenosis, or stenosis caused by traumatic or neoplastic disease, were excluded from the analysis. The approved number of the Institutional Review Board is KBSMC 2023-07-004.
Outcome Measures
On each of the following months after surgery, the 3rd, 6th, and 12th months, clinical and radiologic outcomes were evaluated.
Clinical outcomes are evaluated based on pain and functional perspectives. The level of pain in the back/buttock and leg was determined through a patient-reported outcome method that involved a 10-point visual analog scale (VAS). Additionally, the level of functional disability was measured using the Oswestry Disability Index (ODI), which has been validated for Korean speakers5). The survey was conducted by an unbiased researcher during the follow-up period. The data was collected in a prospective manner and analyzed retrospectively using an image software review and a patient-reported outcome questionnaire in the electronic medical record.
Radiological outcomes were evaluated by conducting serial checks of X-rays, and the parameters were assessed through lateral X-rays in both a neutral and dynamic posture. These parameters were evaluated to determine the progression of spinal instability. Three X-ray parameters were evaluated in detail for assessing possible spinal instability after surgery: (1) neutral slippage percentage, which is the slippage divided by the length of the upper line at the lower vertebra; (2) dynamic slippage percentage, which is the difference in slippage percentage between extension and flexion postures; and (3) dynamic intervertebral angle, which is the difference in the intervertebral angle between extension and flexion postures (Fig. 1). The data were analyzed using the PiView STAR program (INFINITT Healthcare, Seoul, Korea).
Statistical Analysis
Using the mixed-effect model, we compared various parameters such as X-ray measurements, VAS scores for back and buttocks pain, VAS scores for leg pain, and the ODI, recorded at different times for a patient, against baseline values. The random effects considered were the individual patients for clinical outcomes and surgical levels for radiological outcomes. Fixed effects were established as times at baseline and during the postoperative periods. Multiple comparisons between the baseline and each follow-up time value were adjusted using the Bonferroni method.
Data analysis was carried out using IBM SPSS Statistics version 24.0 (IBM Corp., Armonk, NY, USA). Statistically significant results were those with a p-value of less than 0.05.
Surgical Procedure
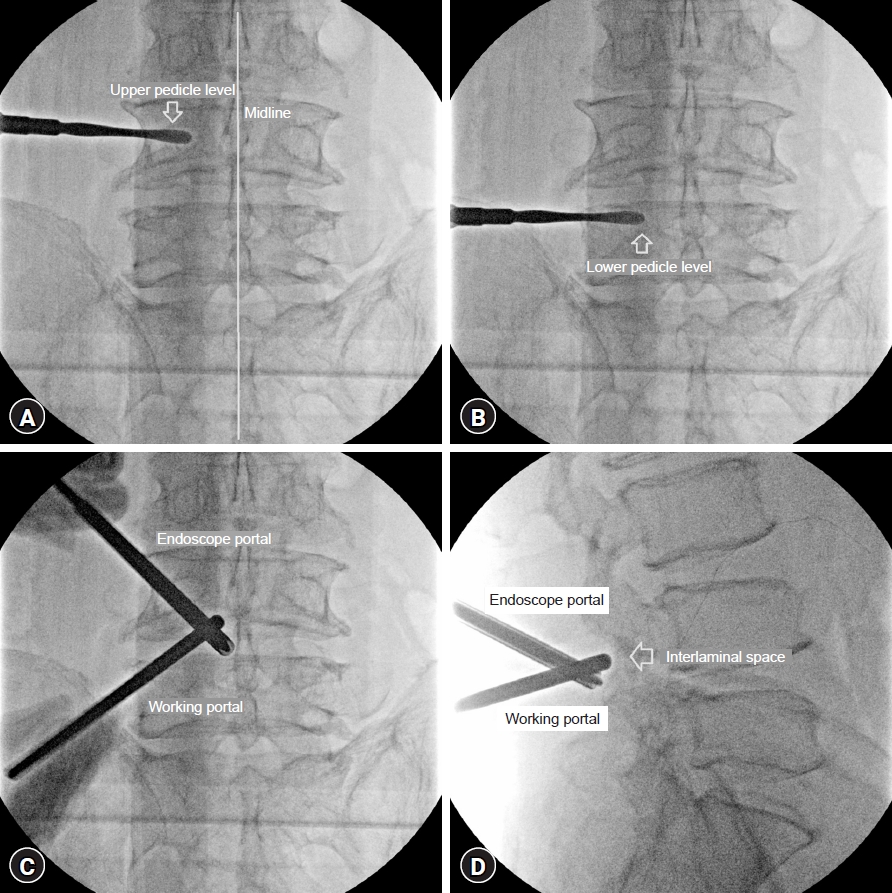
The patient is positioned in a flexed, prone position over a radiolucent frame, and the operation level is confirmed using fluoroscopy. The operation level is ascertained using fluoroscopic guidance. Entry points on the skin are determined based on the location of the pathological lesion and the patient's unique anatomy. Typically, two transverse incisions are made around 1.5 cm above and below the disc space at the edge of the medial interpedicular line (Fig. 2). To facilitate the flow of irrigation fluid, two cross-linear fascial openings, each exceeding 7 mm in length, are created using a 15-blade scalpel. The multifidus muscle is delicately separated using a blunt muscle-splitting approach, employing a series of dilators that touch the lamino-facet junction and a blunt muscle detacher. The accurate placement of the instrument is verified with biplanar fluoroscopic imaging. An endoscope sheath is inserted through one portal for the scope, while a semi-tubular retractor creates a wider working portal. Any minor muscle bleeding is effectively managed using a low-voltage radiofrequency (RF) device.
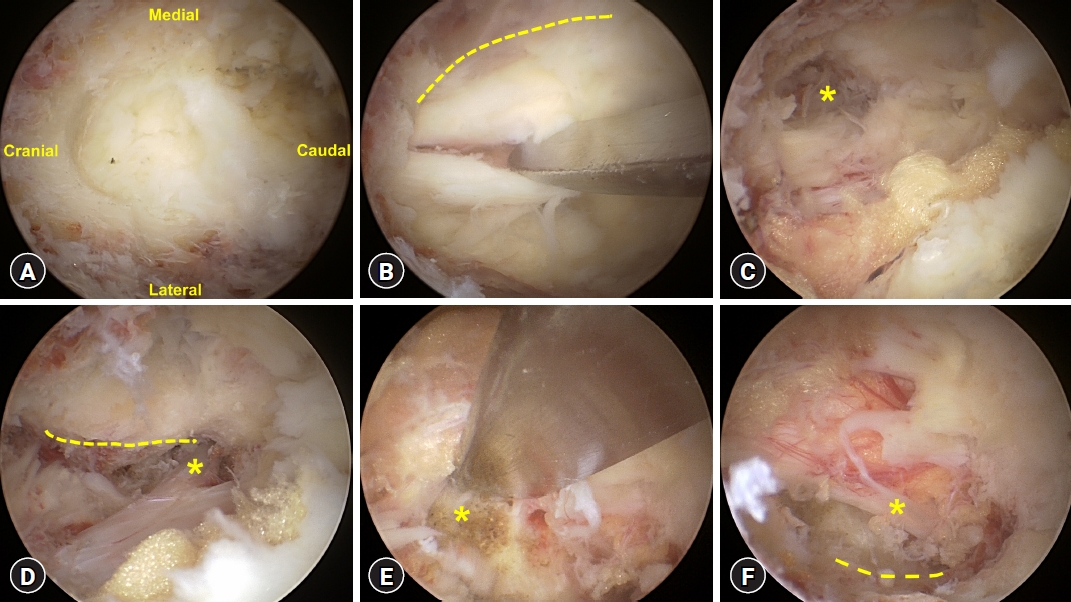
The initial step is ipsilateral decompression at the site of the pathological lesion. Neural structures compressed by hypertrophied facet joints, the lamina, and a thickened ligamentum flavum are selectively and safely removed via drilling. Subsequently, a curette or punch can be used to alleviate the neural structures.
Contralateral decompression can be achieved through an ipsilateral unilateral lamina opening. For this, a wide laminotomy is performed, beginning at the spino-laminar junction and undercutting the base of the spinous process. Partial resection of the spinous process base allows the scope to freely traverse into the contralateral side of the spinal canal. The ligamentum flavum is then separated from the contralateral lamina using a blunt dissector, remaining intact with the dura for the protection of neural structures. To prevent a possible tear of the dura during the procedure, it is important to avoid aggressive removal when there is a strong adhesion between the ligamentum flavum and the dura. Following this, bony decompression of the facet joint and lateral recess is carried out using a 3 mm or 4 mm diamond drill, either cranially or caudally. A partial sublaminar facetectomy is performed to preserve the integrity of the facet joint. Following sufficient bony decompression, the remaining ligamentum flavum is delicately resected using a curette or curved punch to completely relieve the neural structures on both sides (Fig. 3).
During this surgical technique, protecting the nerve during manipulation around a neural structure can be achieved with the assistance of a nerve root retractor held by an assistant. Once bilateral decompression is confirmed using a blunt probe, the surgery is deemed complete. After meticulous control of bleeding using an RF electrode and Flosseal, a 100 cc hemovac is inserted. The instruments and endoscope are then removed, and the skin is closed with a stitch and a sterile strip.
RESULTS
Forty-seven patients were recruited, and the average follow-up period was 9.1 months (interquartile range, 6-12 months). Average age at the time of surgery was 66.5 years (standard deviation [SD], ±10.3). Male to female ratio was 0.57 (17 men and 30 women). Thirty-six patients had single-level decompression, while 11 had two-level decompression. UBE surgery was performed for 58 stenotic level, and L4-L5 level was the most frequent surgical level (40 levels, 69.0%) (Table 1).
No serious complications were noted during the follow-up period. There were 3 additional surgeries after initial UBE surgery during the follow-up. One patient had revision surgery due to a dura tear, and two had revision surgeries due to hematoma.
Significant clinical improvement was obtained after the surgery. Mean preoperative VAS scores of back/buttocks (back VAS), VAS scores of legs (leg VAS), and ODI were 5.2 (SD, ±1.6), 4.9 (SD, ±1.7), and 40.5 (SD, ±14.2) respectively. The 3-month follow-up data (back VAS; 2.3 [SD, ±1.6], leg VAS; 1.9 [SD, ±1.9], ODI; 24.6 [SD, ±15.6]) showed significant improvements compared to the preoperative values (p<0.001, respectively). Mixed-effect model can analyze long-term trend of outcomes after the surgery. The back VAS, leg VAS, and ODI were lowest at 6 months follow-up period, and they showed plateau over the follow-up period (p<0.001, respectively) (Fig. 4).
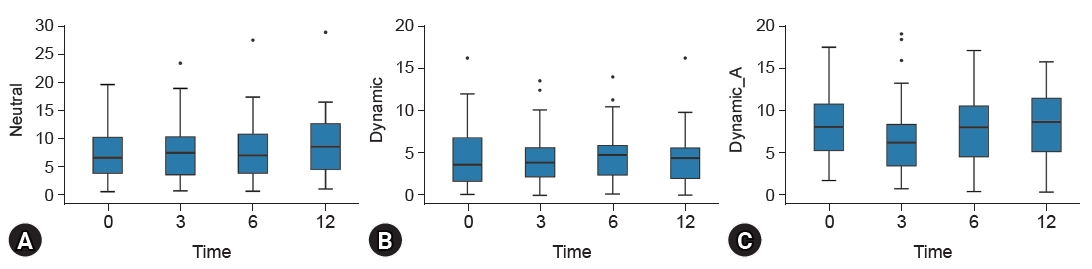
Preoperative average of neutral slippage percentage was 7.7% (SD, ±4.8%), the dynamic slippage percentage (the difference in the slip percentage during flexion and extension posture) was 4.7% (SD, ±3.5%). Another parameter for evaluation of dynamic instability, dynamic intervertebral angle, was checked 8.1° (SD, ± 3.4°) at baseline. During the postoperative period, neutral slippage percentage, dynamic slippage and dynamic intervertebral angle did not progress over the follow-up time, which means favorable maintenance of spinal stability over the follow-up period (Fig. 5).
DISCUSSION
Conventional surgical technique with open laminectomy has long been considered the favored therapeutic approach for LSS. However, extensive soft tissue dissection may lead to paravertebral muscle atrophy and long-term back pain and discomfort3). Over-resection of paraspinal muscles, supraspinous and interspinous ligaments, and facet joints for improved surgical visibility may lead to instability, requiring instrumentation17).
Unilateral laminectomy with bilateral decompression (ULBD) is a surgical technique used to decompress both sides of the spinal canal without having to dissect the contralateral side. It is also used to minimize muscle dissection and has shown satisfactory results2,13). The idea of MI surgery has been utilized in multiple surgical fields and has demonstrated positive outcomes. Several studies have reported satisfactory clinical outcomes of ULBD using microscope11,12). From these points of view, recently, a full endoscopic approach using a uni-portal endoscope has been introduced due to its minimal blood loss, minor complications, and early discharge7). However, this technique is technically difficult to perform and has limited indications due to the narrowed working space within the limited endoscopic channel.
UBE employs a dual portal system - one for the endoscope for viewing and the other for surgical equipment. This technique expands the working space beyond the limited confines of a tubular retractor or inside of an endoscope channel. This allows surgery to be performed from another corridor, allowing surgeons to use their instruments, such as drills, Kerrison rongeurs, curettes, and pituitary forceps, in a manner like open surgeries. It also provides a clear and expansive view of the epidural working space, preserving epidural fat and vessels. Endoscopy offers relatively greater freedom of movement, which allows for easier visualization towards the contralateral ligamentum flavum side and can be advantageous for contralateral decompression. Furthermore, it has a trauma-free working area within the potential adipose space between the multifidus muscles. Unlike other MI procedures that utilize a tubular retractor, this method circumvents crushing and over-retraction injuries surrounding muscles. So, it enables safe and extensive decompression of stenotic lesions while preserving paraspinal muscles and inter- and supra-spinous ligaments for the spinal column's posterior integrity. The present study findings revealed that patients who underwent UBE experienced favorable recovery of back pain, particularly over the early postoperative period. In this study, the degree of improvement in back pain is comparable to the results reported in other studies that investigated other MI surgical techniques, such as microscopic or full-endoscopic surgery6,9). This could be due to the preservation of functioning posterior spinal structures, including muscles and posterior ligaments.
However, surgery using the UBE technique can result in various complications, such as dural tears and epidural hematomas10,14). These complications are maybe linked to the surgical view under water medium, which is different from open microscopic surgery that uses air medium. In UBE surgery, it is crucial to maintain a clear surgical field by controlling epidural bleeding to take advantage of the benefits of a good surgical view while safely and effectively performing decompression. To ensure a clear view, it is important to ensure smooth inflow and outflow of water, and to use meticulous RF and appropriate hemostatic agents. It is also essential to acquire sufficient endoscopic surgical anatomy during the learning curve period.
In this research, the author presents short-term outcomes of UBE for LSS, considering both clinical and radiological parameters. The mixed-effect model helped to account for missing data in repeated measurements, proving useful in retrospectively analyzing this prospectively gathered data during the follow-up. During the follow-up period, there were no significant complications observed. Significant improvements in VAS scores for back/buttock and leg pain were seen postoperatively at 3 months, and these levels were sustained over time (p<0.001). The ODI also saw substantial enhancements immediately after surgery and throughout the follow-up period (p<0.001) without symptom deterioration. These outcomes align with other clinical results from standard or MI ULBD using a microscope12) or uniportal endoscope7).
This observational study had a relatively limited patient pool and a short-term follow-up. To validate these findings, it is vital to conduct long-term prospective controlled trials on a larger scale. These trials should compare the findings with other laminectomy techniques, such as microscopic or uniportal endoscopic ULBD for LSS.