INTRODUCTION
Chordoma is a primary malignant neoplasm that is a slow-growing and locally aggressive tumor3). Although it is relatively rare, chordoma is the most frequent primary malignant tumor in the sacrum. It is found in men more than in women (2:1)11). The sacral chordoma is often large at the time of diagnosis because of its minimal symptoms and deeply seated location.
The first line of treatment is surgical resection rather than chemotherapy or radiation therapy9) because of the poor response to adjuvant therapy. There are three surgical approaches: anterior, posterior and combined. Total en bloc sacrectomy is traditionally performed using the combined anterior-posterior approach. However, the combined approach is surgically demanding with a prolonged operation time and has a longer recovery time2).
The aim of this study is to present our experience with the removal of a huge sacral tumor using a posterior-only approach and to emphasize its feasibility as a practical option.
CASE REPORT
A 57-year-old man with no past medical history presented with a 4-month history of right buttock pain. The patient had paresthesia in the right buttock and S1 and S2 dermatomes without lower extremity motor weakness on neurological examination.
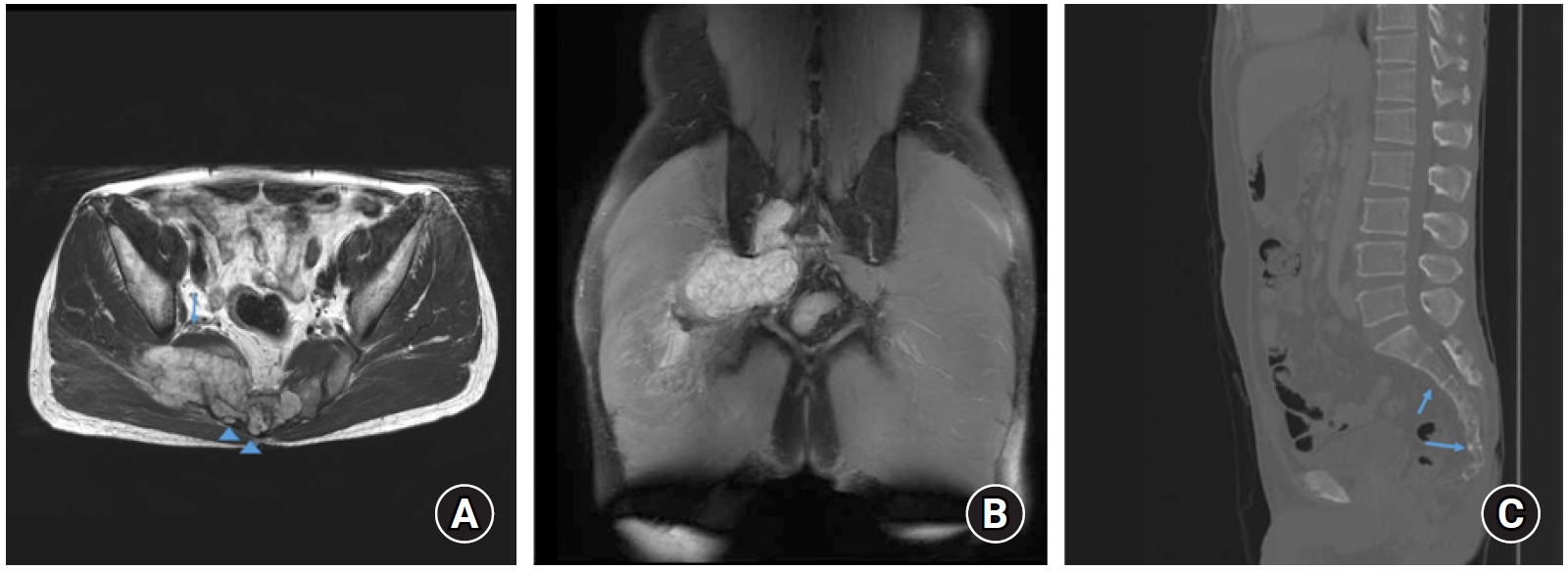
The radiological evaluation of pelvis computed tomographic scan and pelvic bone magnetic resonance imaging, revealed 8.0 × 4.0 × 4.5-cm sized bone destructing mass involving S2 to S5 body and posterior element, which extended dorsally to the right piriformis and gluteal muscles. An extraosseous extension was found in the right paraspinal muscle (Fig.1). It had low signal intensity on T1 and had heterogeneous lobulated mildly increased signal intensity on T2 and contrast-enhanced T1 images. These findings are consistent with those of chordoma1).
Ultrasound-guided gun biopsy was performed for the differential diagnosis of the right pelvis mass. The cells stained positively for epithelial membrane antigen and pan-cytokeratin, and negatively for S-100 (Table 1)10). These findings are compatible with those of chordomas. No intra-abdominal or distant metastases were observed
Surgical Procedure
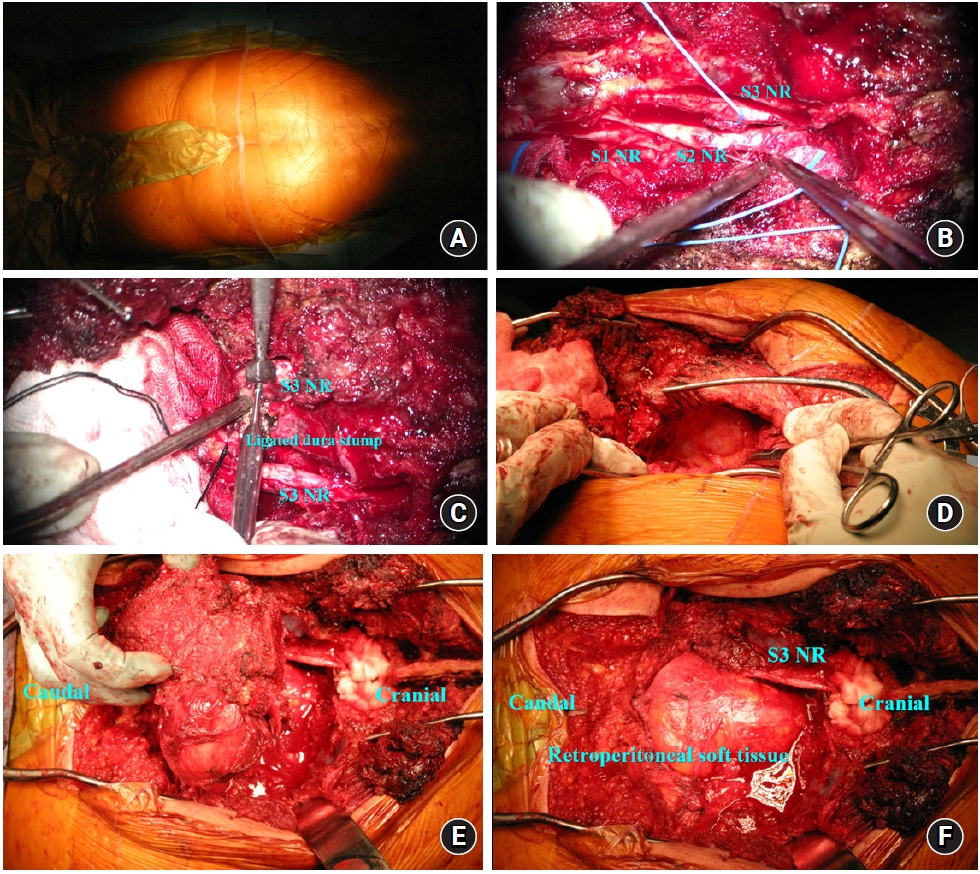
Considering the intracompartmental tumor, we decided to perform a single posterior approach surgery. Under general anesthesia, the patient was flipped to the prone position on a Jackson table. Considering the possibility of a local flap, we prepared a wide drape including both thighs. A midline skin incision L3-sacrococcygeal level was made with a Y-shaped bifurcation direct to bilateral great trochanter (Fig. 2A). Subperiosteal dissection was performed from L3 to the sacroilium, and grayish well-capsulated pudding-like tumors were exposed bilaterally in the bone and neural foramen at the S3 level. A frozen biopsy confirmed chordomas.
For en bloc resection of the tumor, extra-osseous tumor mass around the right piriformis, sacral ala, and gluteus muscle was separated from normal tissue by dissection and cauterization. Then S1 and S2 laminectomy was performed using a bone scalpel and punch. Part of the sacral bone, including the right S2 distal sacral foramen involving the tumor, was also grossly resected using a burr and osteotome.
The sacral resection margin was marked around the S1 and S2 sacral level using drills. An additional fracture was performed on the midline of the sacrum at the S1 and S2 level to separate the fractured sacral bone. The fractured bone was gently removed using a dissector. Sacral nerve roots were identified with bilateral S1 to S2 and left S3 nerve roots preserved (Fig. 2B), and other sacral roots separated from each foramen. The left S3 nerve root was preserved to maintain urinary function (Fig. 2C). The Remaining sacrococcyx was separated from tumor mass from the surrounding pelvic floor structure, pelvic floor anococcygeal ligament, sacrotuberous/sacrospinous ligament, and piriformis muscle (Fig. 2D). Both sacroiliac joints were preserved as much as possible. The femoral attachment of the right piriformis muscle was cut with sacrectomy. A sacrectomy was partially performed with careful blunt dissection of the left side. There was no massive bleeding, vessel or visceral injuries during the procedure. The tumor mass was identified from the bottom of the sacrum to the sacral canal at S3 to S5 and right femur. Finally, en bloc resection was completed via dissection from the periphery (Fig. 2E, 3). The sciatic nerve and left S3 nerve root were intact (Fig.2F)4). No major intraoperative complications occurred.
There was no motor weakness during postoperative care, but mild hypesthesia on the bilateral buttocks developed. Although the patient was able to urinate by himself, residual urine was detected. Adjuvant radiotherapy was administered at the surgical site to prevent local recurrence.
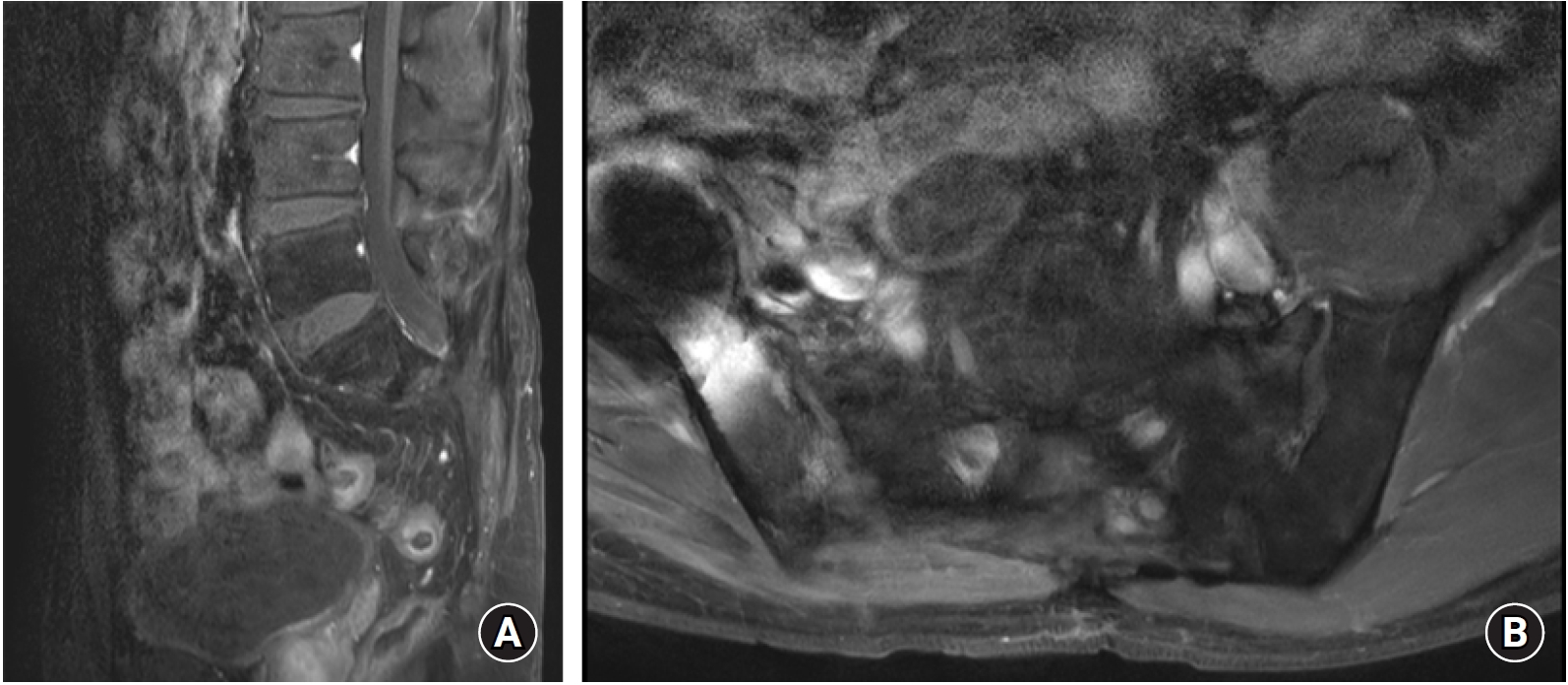
There was no recurrence after five years of follow-up after surgery (Fig.4). The patient did not show postoperative complications including wound healing problems, neurological deficits, or infection.
DISCUSSION
The management of sacral chordoma is quite challenging. Due to the limited response to radiation and chemotherapy, surgical removal of the tumor is the treatment of choice. Several reports have described that sacral chordomas generally present as large masses that are difficult to remove fully. However, a negative resection margin with decreased local recurrences is critical for prognosis12).
Despite recent advances in surgical techniques, sacral tumor resection is associated with several complications. The most commonly reported complications in the literature are massive bleeding during sacrectomy, surgical wound infections, wound dehiscence, neurological deficits (bowel and bladder dysfunction, sciatic nerve or lumbar plexus injury), and instrument-related complications following spinopelvic fixation. Many authors have considered ileostomy and suprapubic bladder catheters unavoidable while operating large chordomas1).
Several studies have been conducted to determine which surgical approaches have a better outcome and fewer complications. In the combined anterior and posterior approach, many complications such as long operation time, a large amount of bleeding, wound dehiscence, and infection has been reported1,6). The posterior only approach was first reported in 19877). Although the posterior-only approach has the disadvantage in dissection of visceral organs and preservation of sacral nerve roots, it has shown favorable results compared to the anterior approach5). The combined approach has revealed the benefit of better resection margin with increased mortality but no increase in long-term survival2,8).
However, the patients in this case who were diagnosed with sacral chordoma involving S2 to S5 sacrum, right piriformis muscle, and gluteal muscles were treated with a single posterior approach, preserving S1, S2, and left S3 nerve roots without significant neurological deficits. It can be a good surgical approach if appropriate case selection is performed with an accurate understanding of the structure of the spine and surrounding structures. The posterior-only approach for the huge sacral tumor presented here can avoid further abdominal visceral injury as well as vessel injury compared to the anterior approach.